| |

| |
| Names | |
|---|---|
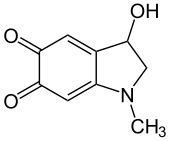
| IUPAC name
3-Hydroxy-1-methyl-2,3-dihydro-1H-indole-5,6-dione
| |
| Other names
Adraxone; Pink adrenaline
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.000.176 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| Properties | |
| C9H9NO3 | |
| Molar mass | 179.175 g·mol−1 |
| Appearance | deep-violet[1] |
| Density | 3.785 g/cm3 |
| Boiling point | 115–120 °C (239–248 °F; 388–393 K) (decomposes) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Adrenochrome is a chemical compound produced by the oxidation of adrenaline (epinephrine). It was the subject of limited research from the 1950s through to the 1970s as a potential cause of schizophrenia. While it has no current medical application, the semicarbazide derivative, carbazochrome, is a hemostatic medication.
Despite this compound's name, it is unrelated to the element chromium; instead, the ‑chrome suffix indicates a relationship to color, as pure adrenochrome has a deep violet coloration.[1]
- ^ a b Heacock RA, Nerenberg C, Payza AN (1 May 1958). "The Chemistry of the "Aminochromes": Part I. The Preparation and Paper Chromatography of Pure Adrenochrome". Canadian Journal of Chemistry. 36 (5): 853–857. doi:10.1139/v58-124.