| Astrocyte | |
|---|---|
 An astrocyte from a rat brain grown in tissue culture and stained with antibodies to GFAP (red) and vimentin (green). Both proteins are present in large amounts in the intermediate filaments of this cell, so the cell appears yellow. The blue material shows DNA visualized with DAPI stain, and reveals the nucleus of the astrocyte and of other cells. Image courtesy of EnCor Biotechnology Inc. | |
| Details | |
| Precursor | Glioblast |
| Location | Brain and spinal cord |
| Identifiers | |
| Latin | astrocytus |
| MeSH | D001253 |
| NeuroLex ID | sao1394521419 |
| TH | H2.00.06.2.00002, H2.00.06.2.01008 |
| FMA | 54537 |
| Anatomical terms of microanatomy | |
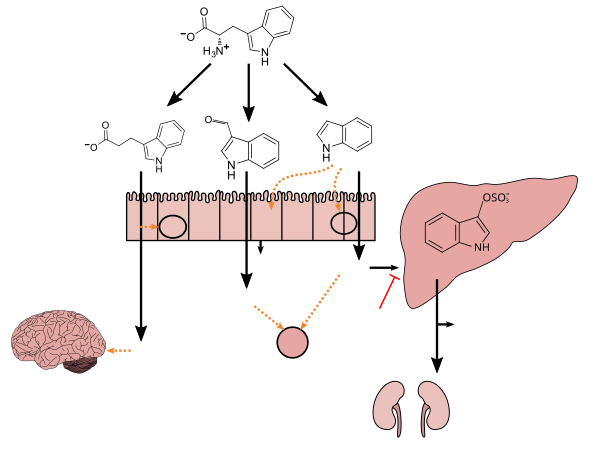
Tryptophan metabolism by human gastrointestinal microbiota ()
|
Astrocytes (from Ancient Greek ἄστρον, ástron, "star" and κύτος, kútos, "cavity", "cell"), also known collectively as astroglia, are characteristic star-shaped glial cells in the brain and spinal cord. They perform many functions, including biochemical control of endothelial cells that form the blood–brain barrier,[5] provision of nutrients to the nervous tissue, maintenance of extracellular ion balance, regulation of cerebral blood flow, and a role in the repair and scarring process of the brain and spinal cord following infection and traumatic injuries.[6] The proportion of astrocytes in the brain is not well defined; depending on the counting technique used, studies have found that the astrocyte proportion varies by region and ranges from 20% to around 40% of all glia.[7] Another study reports that astrocytes are the most numerous cell type in the brain.[6] Astrocytes are the major source of cholesterol in the central nervous system.[8] Apolipoprotein E transports cholesterol from astrocytes to neurons and other glial cells, regulating cell signaling in the brain.[8] Astrocytes in humans are more than twenty times larger than in rodent brains, and make contact with more than ten times the number of synapses.[9]
Research since the mid-1990s has shown that astrocytes propagate intercellular Ca2+ waves over long distances in response to stimulation, and, similar to neurons, release transmitters (called gliotransmitters) in a Ca2+-dependent manner.[10] Data suggest that astrocytes also signal to neurons through Ca2+-dependent release of glutamate.[11] Such discoveries have made astrocytes an important area of research within the field of neuroscience.
- ^ a b c d e f g h i Zhang LS, Davies SS (April 2016). "Microbial metabolism of dietary components to bioactive metabolites: opportunities for new therapeutic interventions". Genome Med. 8 (1): 46. doi:10.1186/s13073-016-0296-x. PMC 4840492. PMID 27102537.
Lactobacillus spp. convert tryptophan to indole-3-aldehyde (I3A) through unidentified enzymes [125]. Clostridium sporogenes convert tryptophan to IPA [6], likely via a tryptophan deaminase. ... IPA also potently scavenges hydroxyl radicals
Table 2: Microbial metabolites: their synthesis, mechanisms of action, and effects on health and disease
Figure 1: Molecular mechanisms of action of indole and its metabolites on host physiology and disease - ^ Wikoff WR, Anfora AT, Liu J, Schultz PG, Lesley SA, Peters EC, Siuzdak G (March 2009). "Metabolomics analysis reveals large effects of gut microflora on mammalian blood metabolites". Proc. Natl. Acad. Sci. U.S.A. 106 (10): 3698–3703. Bibcode:2009PNAS..106.3698W. doi:10.1073/pnas.0812874106. PMC 2656143. PMID 19234110.
Production of IPA was shown to be completely dependent on the presence of gut microflora and could be established by colonization with the bacterium Clostridium sporogenes.
IPA metabolism diagram - ^ "3-Indolepropionic acid". Human Metabolome Database. University of Alberta. Retrieved 12 June 2018.
- ^ Chyan YJ, Poeggeler B, Omar RA, Chain DG, Frangione B, Ghiso J, Pappolla MA (July 1999). "Potent neuroprotective properties against the Alzheimer beta-amyloid by an endogenous melatonin-related indole structure, indole-3-propionic acid". J. Biol. Chem. 274 (31): 21937–21942. doi:10.1074/jbc.274.31.21937. PMID 10419516. S2CID 6630247.
[Indole-3-propionic acid (IPA)] has previously been identified in the plasma and cerebrospinal fluid of humans, but its functions are not known. ... In kinetic competition experiments using free radical-trapping agents, the capacity of IPA to scavenge hydroxyl radicals exceeded that of melatonin, an indoleamine considered to be the most potent naturally occurring scavenger of free radicals. In contrast with other antioxidants, IPA was not converted to reactive intermediates with pro-oxidant activity.
- ^ Suzuki, Yasuhiro; Sa, Qila; Ochiai, Eri; Mullins, Jeremi; Yolken, Robert; Halonen, Sandra K. (2014). "Cerebral Toxoplasmosis". Toxoplasma Gondii. Elsevier. pp. 755–96. doi:10.1016/b978-0-12-396481-6.00023-4. ISBN 978-0-12-396481-6.
Astrocytes are the dominant glial cell in the brain and numerous studies indicate they are central to the intracerebral immune response to T. gondii in the brain.
- ^ a b Freeman, MR; Rowitch, DH (30 October 2013). "Evolving concepts of gliogenesis: a look way back and ahead to the next 25 years". Neuron. 80 (3): 613–23. doi:10.1016/j.neuron.2013.10.034. PMC 5221505. PMID 24183014.
- ^ Verkhratsky A, Butt AM (2013). "Numbers: how many glial cells are in the brain?". Glial Physiology and Pathophysiology. John Wiley and Sons. pp. 93–96. ISBN 978-0-470-97853-5.
- ^ a b Wang, Hao; Kulas, Joshua A.; Ferris, Heather A.; Hansen, Scott B. (2020-10-14). "Regulation of beta-amyloid production in neurons by astrocyte-derived cholesterol". bioRxiv: 2020.06.18.159632. doi:10.1101/2020.06.18.159632. S2CID 220044671.
- ^ Cite error: The named reference
Sloanwas invoked but never defined (see the help page). - ^ "Role of Astrocytes in the Central Nervous System". Retrieved 27 July 2018.
- ^ Fiacco TA, Agulhon C, McCarthy KD (October 2008). "Sorting out astrocyte physiology from pharmacology". Annual Review of Pharmacology and Toxicology. 49 (1): 151–74. doi:10.1146/annurev.pharmtox.011008.145602. PMID 18834310.