| |
| Names | |
|---|---|
| IUPAC name
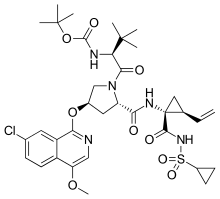
3-Methyl-N-{[(2-methyl-2-propanyl)oxy]carbonyl}-L-valyl-(4R)-4-[(7-chloro-4-methoxy-1-isoquinolinyl)oxy]-N-{(1R,2S)-1-[(cyclopropylsulfonyl)carbamoyl]-2-vinylcyclopropyl}-L-prolinamide
| |
| Systematic IUPAC name
tert-Butyl {(2S)-1-[(32S,34R,61R,62S)-17-chloro-62-ethenyl-14-methoxy-4,7,9,9-tetraoxo-2-oxa-9λ6-thia-5,8-diaza-1(1)-isoquinolina-3(4,2)-pyrrolidina-6(1,1),10(1)-dicyclopropadecaphan-31-yl]-3,3-dimethyl-1-oxobutan-2-yl}carbamate | |
| Other names
BMS-650032
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.206.482 |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C35H46ClN5O9S | |
| Molar mass | 748.29 g·mol−1 |
| Pharmacology | |
| J05AP06 (WHO) | |
| Legal status | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Asunaprevir (formerly BMS-650032, brand name in Japan and Russia[3] Sunvepra) is an experimental drug candidate for the treatment of hepatitis C. It was undergoing development by Bristol-Myers Squibb and has completed Phase III clinical trials in 2013.[4]
Asunaprevir is an inhibitor of the hepatitis C virus enzyme serine protease NS3.[5] Asunaprevir is being tested in combination with pegylated interferon and ribavirin, as well as in interferon-free regimens with other direct-acting antiviral agents including daclatasvir.[6][7][8]
- ^ "Prescription medicines: registration of new chemical entities in Australia, 2015". Therapeutic Goods Administration (TGA). 21 June 2022. Retrieved 10 April 2023.
- ^ "Health Canada New Drug Authorizations: 2016 Highlights". Health Canada. 14 March 2017. Retrieved 7 April 2024.
- ^ "Sunvepra (asunaprevir) soft gelatin capsules 100 mg. Registration certificate". State Register of Medicines (in Russian). Retrieved 26 August 2015.
- ^ "A Phase 3 Study in Combination With BMS-790052 and BMS-650032 in Japanese Hepatitis C Virus (HCV) Patients". ClinicalTrials.gov. 23 September 2015.
- ^ C. Reviriego (2012). "Asunaprevir. HCV serine protein NS3 inhibitor, Treatment of hepatitis C virus". Drugs of the Future. 37 (4): 247–254. doi:10.1358/dof.2012.037.04.1789350.
- ^ Lok AS, Gardiner DF, Lawitz E, Martorell C, Everson GT, Ghalib R, et al. (January 2012). "Preliminary study of two antiviral agents for hepatitis C genotype 1". The New England Journal of Medicine. 366 (3): 216–24. doi:10.1056/NEJMoa1104430. PMID 22256805.
- ^ "Bristol-Myers' Daclatasvir, Asunaprevir Cured 77%: Study". Bloomberg. 19 April 2012.
- ^ AASLD: Daclatasvir plus Asunaprevir Rapidly Suppresses HCV in Prior Null Responders Archived 2015-02-08 at the Wayback Machine. Highleyman, L. HIVandHepatitis.com. 8 November 2011.