The Bamberger rearrangement is the chemical reaction of phenylhydroxylamines with strong aqueous acid, which will rearrange to give 4-aminophenols.[1] It is named for the German chemist Eugen Bamberger (1857–1932).[2][3]
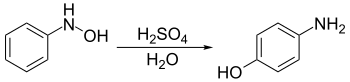
The starting phenylhydroxylamines are typically synthesized by the transfer hydrogenation of nitrobenzenes using rhodium[4] or zinc[5] catalysts.
One application is in the synthesis of fenhexamid.
- ^ Harman, R. E. (1955). "Chloro-p-benzoquinone" (PDF). Organic Syntheses. 35: 22; Collected Volumes, vol. 4, p. 148.
- ^ Bamberger, E. (1894). "Ueber die Reduction der Nitroverbindungen". Chemische Berichte. 27 (2): 1347–1350. doi:10.1002/cber.18940270229.
- ^ Bamberger, E. (1894). "Ueber das Phenylhydroxylamin". Chemische Berichte. 27 (2): 1548–1557. doi:10.1002/cber.18940270276.
- ^ Oxley, P. W.; Adger, B. M.; Sasse, M. J.; Forth1, M. A. (1989). "N-Acetyl-N-Phenylhydroxylamine Via Catalytic Transfer Hydrogenation of Nitrobenzene Using Hydrazine and Rhodium on Carbon". Organic Syntheses. 67: 187. doi:10.15227/orgsyn.067.0187.
{{cite journal}}: CS1 maint: numeric names: authors list (link) - ^ Kamm, O. (1925). "β-Phenylhydroxylamine". Organic Syntheses. 4: 57; Collected Volumes, vol. 1, p. 445. (download PDF)