| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| BAs | |
| Molar mass | 85.733 g/mol[1] |
| Appearance | Brown cubic crystals[1] |
| Density | 5.22 g/cm3[1] |
| Melting point | 1,100 °C (2,010 °F; 1,370 K) decomposes[1] |
| Insoluble | |
| Band gap | 1.82 eV |
| Thermal conductivity | 1300 W/(m·K) (300 K) |
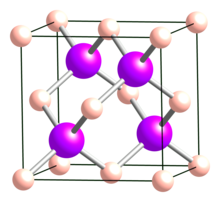
| Structure[2] | |
| Cubic (sphalerite), cF8, No. 216 | |
| F43m | |
a = 0.4777 nm
| |
Formula units (Z)
|
4 |
| Related compounds | |
Other anions
|
Boron nitride Boron phosphide Boron antimonide |
Other cations
|
Aluminium arsenide Gallium arsenide Indium arsenide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |

| |
| Identifiers | |
|---|---|
| Properties | |
| B12As2 | |
| Molar mass | 279.58 g/mol |
| Density | 3.56 g/cm3[3] |
| Insoluble | |
| Band gap | 3.47 eV |
| Structure[4] | |
| Rhombohedral, hR42, No. 166 | |
| R3m | |
a = 0.6149 nm, b = 0.6149 nm, c = 1.1914 nm α = 90°, β = 90°, γ = 120°
| |
Formula units (Z)
|
6 |
| Related compounds | |
Other anions
|
Boron suboxide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Boron arsenide (or Arsenic boride) is a chemical compound involving boron and arsenic, usually with a chemical formula BAs. Other boron arsenide compounds are known, such as the subarsenide B12As2. Chemical synthesis of cubic BAs is very challenging and its single crystal forms usually have defects.
- ^ a b c d Haynes, William M., ed. (2011). CRC Handbook of Chemistry and Physics (92nd ed.). Boca Raton, FL: CRC Press. p. 4.53. ISBN 1-4398-5511-0.
- ^ Perri, J. A; La Placa, S; Post, B (1958). "New group III-group V compounds: BP and BAs". Acta Crystallographica. 11 (4): 310. doi:10.1107/S0365110X58000827.
- ^ Villars, Pierre (ed.) "B12As2 (B6As) Crystal Structure" in Inorganic Solid Phases, Springer, Heidelberg (ed.) SpringerMaterials
- ^ Morosin, B; Aselage, T. L; Feigelson, R. S (2011). "Crystal Structure Refinements of Rhombohedral Symmetry Materials Containing Boron-Rich Icosahedra". MRS Proceedings. 97. doi:10.1557/PROC-97-145.