| |||
| |||
| Names | |||
|---|---|---|---|
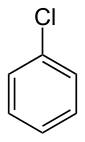
| Preferred IUPAC name
Chlorobenzene | |||
| Other names
Phenyl chloride, monochlorobenzene[1]
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 3DMet | |||
| Abbreviations | PhCl | ||
| 605632 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.003.299 | ||
| EC Number |
| ||
| 26704 | |||
| KEGG | |||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1134 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C6H5Cl | |||
| Molar mass | 112.56 g/mol | ||
| Appearance | colorless liquid | ||
| Odor | almond-like[2] | ||
| Density | 1.11 g/cm3, liquid | ||
| Melting point | −45.58 °C (−50.04 °F; 227.57 K) | ||
| Boiling point | 131.70 °C (269.06 °F; 404.85 K) | ||
| 0.5 g l−1 in water at 20 °C | |||
| Solubility in other solvents | soluble in most organic solvents | ||
| Vapor pressure | 9 mmHg[2] | ||
| −69.97·10−6 cm3/mol | |||
Refractive index (nD)
|
1.52138 | ||
| Viscosity | 0.7232 | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Low to moderate hazard[3] | ||
| GHS labelling: | |||
  
| |||
| Warning | |||
| H226, H302, H305, H315, H332, H411 | |||
| P210, P233, P240, P241, P242, P243, P261, P264, P271, P273, P280, P302+P352, P303+P361+P353, P304+P312, P304+P340, P312, P321, P332+P313, P362, P370+P378, P391, P403+P235, P501 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 29 °C (84 °F; 302 K) | ||
| Explosive limits | 1.3%-9.6%[2] | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
2290 mg/kg (rat, oral)
590 mg/kg (mouse, orally)[4] | ||
LCLo (lowest published)
|
8000 ppm (cat, 3 hr)[5] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 75 ppm (350 mg/m3)[2] | ||
REL (Recommended)
|
none[2] | ||
IDLH (Immediate danger)
|
1000 ppm[2] | ||
| Related compounds | |||
Related Halobenzenes
|
Fluorobenzene Bromobenzene Iodobenzene | ||
Related compounds
|
benzene 1,4-dichlorobenzene | ||
| Supplementary data page | |||
| Chlorobenzene (data page) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Chlorobenzene (abbreviated PhCl) is an aryl chloride and the simplest of the chlorobenzenes, consisting of a benzene ring substituted with one chlorine atom. Its chemical formula is C6H5Cl. This colorless, flammable liquid is a common solvent and a widely used intermediate in the manufacture of other chemicals.[6]
- ^ Pubchem. "Chlorobenzene". pubchem.ncbi.nlm.nih.gov. Retrieved 2022-08-21.
- ^ a b c d e f NIOSH Pocket Guide to Chemical Hazards. "#0121". National Institute for Occupational Safety and Health (NIOSH).
- ^ Chlorobenzene toxicity
- ^ Chlorobenzene: LD50
- ^ a b "Chlorobenzene". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ Cite error: The named reference
ClPhwas invoked but never defined (see the help page).