| |
| Names | |
|---|---|
| IUPAC name
Chromium(III) iodide
| |
| Other names
Chromium triiodide, chromic iodide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.033.614 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| CrI3 | |
| Molar mass | 432.7095 g·mol−1 |
| Appearance | black solid |
| Density | 5.32 g/cm3[1] |
| Melting point | > 600 °C (1,112 °F; 873 K) |
| Soluble | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Chromium(III) iodide, also known as chromium triiodide, is an inorganic compound with the formula CrI3. It is a black solid that is used to prepare other chromium iodides.[2]
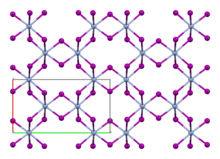
Like the isomorphous chromium(III) chloride (CrCl3), chromium(III) iodide exhibits a cubic-closest packing arrangement in a double-layer crystal lattice. In this structure, chromium exhibits octahedral coordination geometry.[3]
- ^ Perry, Dale L. (2011). Handbook of Inorganic Compounds, Second Edition. Boca Raton, Florida: CRC Press. p. 123. ISBN 978-1-43981462-8. Retrieved 2014-01-10.
- ^ Gregory, N. W.; Handy, L. L. (1957). "Chromium (III) Iodide". Inorganic Syntheses. Vol. 5. pp. 128–130. doi:10.1002/9780470132364.ch34.
{{cite book}}:|journal=ignored (help) - ^ Gregory, N. W.; Handy, L. L. (1952). "Structural Properties of Chromium(III) Iodide and Some Chromium(III) Mixed Halides". J. Am. Chem. Soc. 74 (4): 891–893. doi:10.1021/ja01124a009.