| |

| |
| Names | |
|---|---|
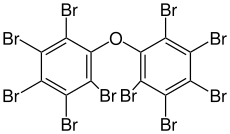
| Preferred IUPAC name
1,1′-Oxybis(2,3,4,5,6-pentabromobenzene) | |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.013.277 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C12Br10O | |
| Molar mass | 959.17 g/mol |
| Appearance | White or pale yellow solid |
| Density | 3.364 g/cm3 solid |
| Melting point | 294 to 296 °C (561 to 565 °F; 567 to 569 K)[1] |
| Boiling point | 425 °C (797 °F; 698 K) (decomposition)[1] |
| 20-30 μg/litre [2] | |
| Hazards | |
| GHS labelling: | |
 
| |
| Warning | |
| H302, H312, H319, H332, H341, H373, H413 | |
| P201, P202, P260, P261, P264, P270, P271, P273, P280, P281, P301+P312, P302+P352, P304+P312, P304+P340, P305+P351+P338, P308+P313, P312, P314, P322, P330, P337+P313, P363, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | 241 °C (466 °F; 514 K) |
| Safety data sheet (SDS) | [1] |
| Related compounds | |
Related polybrominated diphenyl ethers
|
pentabromodiphenyl ether, octabromodiphenyl ether |
Related compounds
|
Diphenyl ether |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Decabromodiphenyl ether (also referred to as decaBDE, DBDE, BDE-209) is a brominated flame retardant which belongs to the group of polybrominated diphenyl ethers (PBDEs). It was commercialised in the 1970s and was initially thought to be safe,[3][4] but is now recognised as a hazardous and persistent pollutant. It was added to Annex A of the Stockholm Convention on Persistent Organic Pollutants in 2017,[5][6] which means that treaty members must take measures to eliminate its production and use. The plastics industry started switching to decabromodiphenyl ethane as an alternative in the 1990s, but this is now also coming under regulatory pressure due to concerns over human health.
- ^ a b Record of Decabromodiphenyl ether in the GESTIS Substance Database of the Institute for Occupational Safety and Health, accessed on 17 June 2008.
- ^ "Environmental Health Criteria". ICPS. Retrieved 2009-07-20.
- ^ Liepins, R; Pearce, E M (October 1976). "Chemistry and toxicity of flame retardants for plastics". Environmental Health Perspectives. 17: 55–63. doi:10.1289/ehp.761755. PMC 1475265. PMID 1026419.
- ^ Norris, J M; Kociba, R J; Schwetz, B A; Rose, J Q; Humiston, C G; Jewett, G L; Gehring, P J; Mailhes, J B (June 1975). "Toxicology of octabromobiphenyl and decabromodiphenyl oxide". Environmental Health Perspectives. 11: 153–161. doi:10.1289/ehp.7511153. PMC 1475203. PMID 126149.
- ^ "c-decaBDE". chm.pops.int. Secretariat of the Stockholm Convention. Retrieved 8 January 2023.
- ^ Reference: C.N.766.2017.TREATIES-XXVII.15 (Depositary Notification)
