 | |
| Clinical data | |
|---|---|
| Trade names | Tikosyn |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601235 |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 96% (oral) |
| Protein binding | 60% -70% |
| Elimination half-life | 10 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.166.441 |
| Chemical and physical data | |
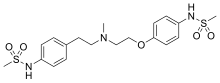
| Formula | C19H27N3O5S2 |
| Molar mass | 441.56 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Dofetilide is a class III antiarrhythmic agent.[2] It is marketed under the trade name Tikosyn by Pfizer, and is available in the United States in capsules containing 125, 250, and 500 μg of dofetilide. It is not available in Europe[3] or Australia.[4]
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 Oct 2023.
- ^ Lenz TL, Hilleman DE (July 2000). "Dofetilide, a new class III antiarrhythmic agent". Pharmacotherapy. 20 (7): 776–786. doi:10.1592/phco.20.9.776.35208. PMID 10907968. S2CID 19897963.
- ^ Wathion N (2004-04-13). "Public Statement on Tikosyn (dofetilide): Voluntary Withdrawal of the Marketing Authorisation in the European Union" (PDF). European Agency for the Evaluation of Medicinal Products. Archived from the original (PDF) on 2017-10-13. Retrieved 2015-01-03.
- ^ Australian Medicines Handbook 2014