| |

| |

| |
| Names | |
|---|---|
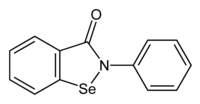
| Preferred IUPAC name
2-Phenyl-1,2-benzoselenazol-3(2H)-one | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.132.190 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C13H9NOSe | |
| Molar mass | 274.17666 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Ebselen (also called PZ 51, DR3305, and SPI-1005), is a synthetic organoselenium drug molecule with anti-inflammatory, anti-oxidant and cytoprotective activity. It acts as a mimic of glutathione peroxidase and can also react with peroxynitrite.[1] It is being investigated as a possible treatment for reperfusion injury and stroke,[2][3] Ménière's disease,[4][5] hearing loss and tinnitus,[6][7] and bipolar disorder.[8][9]
Additionally, ebselen may be effective against Clostridioides difficile infections[10] and has been shown to have antifungal activity against Aspergillus fumigatus.[11]
Ebselen is a potent scavenger of hydrogen peroxide as well as hydroperoxides including membrane bound phospholipid and cholesterylester hydroperoxides. Several ebselen analogs have been shown to scavenge hydrogen peroxide in the presence of thiols.[12]
- ^ Schewe T (October 1995). "Molecular actions of ebselen--an antiinflammatory antioxidant". General Pharmacology. 26 (6): 1153–69. doi:10.1016/0306-3623(95)00003-J. PMID 7590103.
- ^ Parnham M, Sies H (March 2000). "Ebselen: prospective therapy for cerebral ischaemia". Expert Opinion on Investigational Drugs. 9 (3): 607–19. doi:10.1517/13543784.9.3.607. PMID 11060699. S2CID 42599736.
- ^ Yamaguchi T, Sano K, Takakura K, Saito I, Shinohara Y, Asano T, Yasuhara H (January 1998). "Ebselen in acute ischemic stroke: a placebo-controlled, double-blind clinical trial. Ebselen Study Group". Stroke. 29 (1): 12–7. doi:10.1161/01.STR.29.1.12. PMID 9445321.
- ^ "SPI-1005 for the Treatment of Meniere's Disease". Sound Pharmaceuticals. Retrieved 7 November 2022.
- ^ "Sound Pharma initiates enrollment in pivotal Phase 3 clinical trial of SPI-1005 for the treatment of hearing loss and tinnitus in Meniere's disease". Sound Pharmaceuticals. 2022-07-28. Retrieved 7 November 2022.
- ^ Kil J, Pierce C, Tran H, Gu R, Lynch ED (April 2007). "Ebselen treatment reduces noise induced hearing loss via the mimicry and induction of glutathione peroxidase". Hearing Research. 226 (1–2): 44–51. doi:10.1016/j.heares.2006.08.006. PMID 17030476. S2CID 39896274.
- ^ Kil, Jonathan; Harruff, E. Emily; Longenecker, Ryan J. (2022). "Development of ebselen for the treatment of sensorineural hearing loss and tinnitus". Hearing Research. 413. Elsevier BV: 108209. doi:10.1016/j.heares.2021.108209. ISSN 0378-5955. PMID 33678494. S2CID 231956488.
- ^ Singh N, Halliday AC, Thomas JM, Kuznetsova OV, Baldwin R, Woon EC, et al. (2013). "A safe lithium mimetic for bipolar disorder". Nature Communications. 4: 1332. Bibcode:2013NatCo...4.1332S. doi:10.1038/ncomms2320. PMC 3605789. PMID 23299882.
- ^ "New drug for bipolar disorder may offer fewer side effects". University of Oxford Latest News. 2013-01-08. Retrieved 12 January 2013.
- ^ "Drug disarms deadly C. difficile bacteria without destroying healthy gut flora". Medical Express.
- ^ Marshall AC, Kidd SE, Lamont-Friedrich SJ, Arentz G, Hoffmann P, Coad BR, Bruning JB (March 2019). "Structure, Mechanism, and Inhibition of Aspergillus fumigatus Thioredoxin Reductase". Antimicrobial Agents and Chemotherapy. 63 (3): e02281-18. doi:10.1128/AAC.02281-18. PMC 6395915. PMID 30642940.
- ^ Satheeshkumar K, Mugesh G (April 2011). "Synthesis and antioxidant activity of peptide-based ebselen analogues". Chemistry: A European Journal. 17 (17): 4849–57. doi:10.1002/chem.201003417. PMID 21400619.