 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Emcyt, Estracyt |
| Other names | EMP; Leo 299; NSC-89199; Ro 21-8837/001; Estradiol normustine phosphate; Estradiol 3-normustine 17β-phosphate; Estradiol 3-(bis(2-chloroethyl)carbamate) 17β-(dihydrogen phosphate) |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a608046 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth, IV |
| Drug class | Chemotherapeutic agent; Estrogen; Estrogen ester |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 44–75% (as estramustine and estromustine)[1] |
| Protein binding | • Estradiol: 98%[2] • Estrone: 96%[2] |
| Metabolism | Liver, intestines[3][1][6] |
| Metabolites | • Estramustine[3][1] • Estromustine[3][1] • Estradiol[3][1] • Estrone[3][1] • Phosphoric acid[3][1] • Normustine[4] |
| Elimination half-life | • EMP: 1.27 hours[5] • Estromustine: 10–14 hrs[1] • Estrone: 15–17 hours[1] |
| Excretion | Bile, feces (2.9–4.8%)[1][6] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.023.193 |
| Chemical and physical data | |
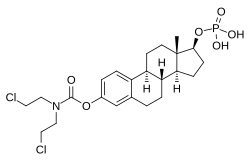
| Formula | C23H32Cl2NO6P |
| Molar mass | 520.38 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Estramustine phosphate (EMP), also known as estradiol normustine phosphate and sold under the brand names Emcyt and Estracyt, is a dual estrogen and chemotherapy medication which is used in the treatment of prostate cancer in men.[7][4][8][9][10][3][1][11][5][12] It is taken multiple times a day by mouth or by injection into a vein.[7][8][3][1][5][12]
Side effects of EMP include nausea, vomiting, gynecomastia, feminization, demasculinization, sexual dysfunction, blood clots, and cardiovascular complications.[3][9][13] EMP is a dual cytostatic and hence chemotherapeutic agent and a hormonal anticancer agent of the estrogen type.[1][3][14][5] It is a prodrug of estramustine and estromustine in terms of its cytostatic effects and a prodrug of estradiol in relation to its estrogenic effects.[1][3] EMP has strong estrogenic effects at typical clinical dosages, and consequently has marked antigonadotropic and functional antiandrogenic effects.[4][1][3][14]
EMP was introduced for medical use in the early 1970s.[3] It is available in the United States, Canada, the United Kingdom, other European countries, and elsewhere in the world.[15][16]
- ^ a b c d e f g h i j k l m n o Bergenheim AT, Henriksson R (February 1998). "Pharmacokinetics and pharmacodynamics of estramustine phosphate". Clinical Pharmacokinetics. 34 (2): 163–172. doi:10.2165/00003088-199834020-00004. PMID 9515186. S2CID 1943973.
- ^ a b Cite error: The named reference
pmid16112947was invoked but never defined (see the help page). - ^ a b c d e f g h i j k l m Kitamura T (February 2001). "Necessity of re-evaluation of estramustine phosphate sodium (EMP) as a treatment option for first-line monotherapy in advanced prostate cancer". International Journal of Urology. 8 (2): 33–36. doi:10.1046/j.1442-2042.2001.00254.x. PMID 11240822. S2CID 43384150.
- ^ a b c Inoue T (2018). "Role of Estramustine Phosphate and Other Estrogens for Castration-Resistant Prostate Cancer". Hormone Therapy and Castration Resistance of Prostate Cancer. Springer. pp. 249–256. doi:10.1007/978-981-10-7013-6_26. ISBN 978-981-10-7012-9.
- ^ a b c d Perry CM, McTavish D (July 1995). "Estramustine phosphate sodium. A review of its pharmacodynamic and pharmacokinetic properties, and therapeutic efficacy in prostate cancer". Drugs & Aging. 7 (1): 49–74. doi:10.2165/00002512-199507010-00006. PMID 7579781. S2CID 36725810.
- ^ a b "Emcyt (estramustine) dosing, indications, interactions, adverse effects, and more". Medscape Reference. WebMD. Retrieved 8 February 2014.
- ^ a b "Emcyt estramustine phosphate sodium capsules" (PDF). FDA. Retrieved 14 March 2022.
- ^ a b Qin Z, Li X, Zhang J, Tang J, Han P, Xu Z, et al. (September 2016). "Chemotherapy with or without estramustine for treatment of castration-resistant prostate cancer: A systematic review and meta-analysis". Medicine. 95 (39): e4801. doi:10.1097/MD.0000000000004801. PMC 5265899. PMID 27684806.
- ^ a b Ravery V, Fizazi K, Oudard S, Drouet L, Eymard JC, Culine S, et al. (December 2011). "The use of estramustine phosphate in the modern management of advanced prostate cancer". BJU International. 108 (11): 1782–1786. doi:10.1111/j.1464-410X.2011.10201.x. PMID 21756277. S2CID 33456591.
- ^ Simpson D, Wagstaff AJ (2003). "Estramustine Phosphate Sodium". American Journal of Cancer. 2 (5): 373–390. doi:10.2165/00024669-200302050-00013. S2CID 70507026.
- ^ Hudes G (February 1997). "Estramustine-based chemotherapy". Seminars in Urologic Oncology. 15 (1): 13–19. PMID 9050135.
- ^ a b Kreis W (1995). "Estramustine revisited". Concepts, Mechanisms, and New Targets for Chemotherapy. Cancer Treatment and Research. Vol. 78. Springer. pp. 163–184. doi:10.1007/978-1-4615-2007-8_8. ISBN 978-1-4613-5829-9. PMID 8595142.
- ^ Cite error: The named reference
pmid9274433was invoked but never defined (see the help page). - ^ a b Cite error: The named reference
OettelSchillinger2012was invoked but never defined (see the help page). - ^ Cite error: The named reference
IndexNominum2000was invoked but never defined (see the help page). - ^ Cite error: The named reference
Drugs.comwas invoked but never defined (see the help page).