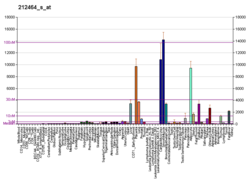
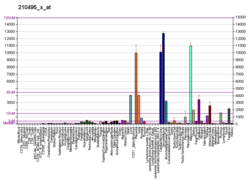
Fibronectin is a high-molecular weight (~500-~600 kDa)[5] glycoprotein of the extracellular matrix that binds to membrane-spanning receptor proteins called integrins.[6] Fibronectin also binds to other extracellular matrix proteins such as collagen, fibrin, and heparan sulfate proteoglycans (e.g. syndecans).
Fibronectin exists as a protein dimer, consisting of two nearly identical monomers linked by a pair of disulfide bonds.[6] The fibronectin protein is produced from a single gene, but alternative splicing of its pre-mRNA leads to the creation of several isoforms.
Two types of fibronectin are present in vertebrates:[6]
- soluble plasma fibronectin (formerly called "cold-insoluble globulin", or CIg) is a major protein component of blood plasma (300 μg/ml) and is produced in the liver by hepatocytes.
- insoluble cellular fibronectin is a major component of the extracellular matrix. It is secreted by various cells, primarily fibroblasts, as a soluble protein dimer and is then assembled into an insoluble matrix in a complex cell-mediated process.
Fibronectin plays a major role in cell adhesion, growth, migration, and differentiation, and it is important for processes such as wound healing and embryonic development.[6] Altered fibronectin expression, degradation, and organization has been associated with a number of pathologies, including cancer, arthritis, and fibrosis.[7][8]
- ^ a b c GRCh38: Ensembl release 89: ENSG00000115414 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000026193 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Mitrović S, Mitrović D, Todorović V (July 1995). "[Fibronectin--a multifunctional glycoprotein]". Srpski Arhiv Za Celokupno Lekarstvo. 123 (7–8): 198–201. PMID 17974429. Archived from the original on February 9, 2022.
- ^ a b c d Pankov R, Yamada KM (October 2002). "Fibronectin at a glance". Journal of Cell Science. 115 (Pt 20): 3861–3. doi:10.1242/jcs.00059. PMID 12244123.
- ^ Williams CM, Engler AJ, Slone RD, Galante LL, Schwarzbauer JE (May 2008). "Fibronectin expression modulates mammary epithelial cell proliferation during acinar differentiation". Cancer Research. 68 (9): 3185–92. doi:10.1158/0008-5472.CAN-07-2673. PMC 2748963. PMID 18451144.
- ^ Kragstrup TW, Sohn DH, Lepus CM, Onuma K, Wang Q, Robinson WH, et al. (2019). "Fibroblast-like synovial cell production of extra domain A fibronectin associates with inflammation in osteoarthritis". BMC Rheumatology. 3: 46. doi:10.1186/s41927-019-0093-4. PMC 6886182. PMID 31819923.