| |

| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
9H-Fluorene[2] | |
| Systematic IUPAC name
Tricyclo[7.4.0.02,7]trideca-2,4,6,9,11,13-hexaene | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.001.541 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C13H10 | |
| Molar mass | 166.223 g·mol−1 |
| Density | 1.202 g/mL |
| Melting point | 116 to 117 °C (241 to 243 °F; 389 to 390 K) |
| Boiling point | 295 °C (563 °F; 568 K) |
| 1.992 mg/L | |
| Solubility | organic solvents |
| log P | 4.18 |
| Acidity (pKa) | 22.6 |
| -110.5·10−6 cm3/mol | |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | 152 °C (306 °F; 425 K) |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
16000 mg/kg (oral, rat) |
| Safety data sheet (SDS) | Sigma-Aldrich |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
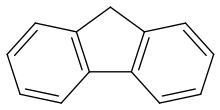
Fluorene /ˈflʊəriːn/, or 9H-fluorene is an organic compound with the formula (C6H4)2CH2. It forms white crystals that exhibit a characteristic, aromatic odor similar to that of naphthalene. Despite its name, it does not contain the element fluorine, but rather it comes from the violet fluorescence it exhibits. For commercial purposes it is obtained from coal tar,[3] where it was discovered and named by Marcellin Berthelot in 1867.[4]
It is insoluble in water and soluble in many organic solvents. Although sometimes classified as a polycyclic aromatic hydrocarbon, the five-membered ring has no aromatic properties.[citation needed] Fluorene is mildly acidic.
- ^ Merck Index, 11th Edition, 4081
- ^ International Union of Pure and Applied Chemistry (2014). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013. The Royal Society of Chemistry. p. 207. doi:10.1039/9781849733069. ISBN 978-0-85404-182-4.
- ^ Cite error: The named reference
Ullmannswas invoked but never defined (see the help page). - ^ Annales de chimie et de physique (in French). Masson. 1867.
