| |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Fluoroacetic acid | |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| 3DMet | |
| 1739053 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.005.120 |
| EC Number |
|
| 25730 | |
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 2642 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
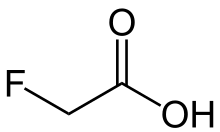
| FCH2CO2H | |
| Molar mass | 78.042 g·mol−1 |
| Appearance | White solid |
| Density | 1.369 g/cm3 |
| Melting point | 35.2 °C (95.4 °F; 308.3 K) |
| Boiling point | 165 °C (329 °F; 438 K) |
| Soluble in water and ethanol | |
| Acidity (pKa) | 2.586[contradictory] |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Highly toxic and corrosive |
| GHS labelling: | |
  
| |
| Danger | |
| H300, H314, H400 | |
| P260, P264, P270, P273, P280, P301+P310, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P330, P363, P391, P405, P501 | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
7 mg/kg (rat, oral) |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Fluoroacetic acid is a organofluorine compound with the chemical formula FCH2CO2H. It is a colorless solid that is noted for its relatively high toxicity.[1] The conjugate base, fluoroacetate occurs naturally in at least 40 plants in Australia, Brazil, and Africa. It is one of only five known organofluorine-containing natural products.[2]
- ^ Timperley, Christopher M. (2000). "Highly-toxic fluorine compounds". Fluorine Chemistry at the Millennium. pp. 499–538. doi:10.1016/B978-008043405-6/50040-2. ISBN 9780080434056.
- ^ K.K. Jason Chan; David O'Hagan (2012). "The Rare Fluorinated Natural Products and Biotechnological Prospects for Fluorine Enzymology". Natural Product Biosynthesis by Microorganisms and Plants, Part B. Methods in Enzymology. Vol. 516. pp. 219–235. doi:10.1016/B978-0-12-394291-3.00003-4. ISBN 9780123942913. PMID 23034231.