 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Emend, Ivemend |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| MedlinePlus | a604003 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Intravenous |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | n/a |
| Protein binding | >95% (aprepitant) |
| Metabolism | To aprepitant |
| Elimination half-life | 9 to 13 hours (aprepitant) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
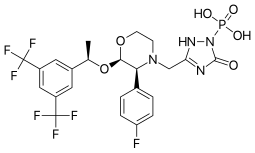
| Formula | C23H22F7N4O6P |
| Molar mass | 614.414 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Fosaprepitant, sold under the brand names Emend (US) and Ivemend (EU) among others, is an antiemetic medication,[6] administered intravenously. It is a prodrug of aprepitant.
Fosaprepitant was developed by Merck & Co. and was approved for medical use in the United States,[7] and in the European Union in January 2008.[5]
- ^ "Prescribing medicines in pregnancy database". Therapeutic Goods Administration (TGA). 21 June 2022. Archived from the original on 2 March 2023. Retrieved 28 April 2023.
- ^ "Emend IV fosaprepitant 150mg (as fosaprepitant dimeglumine) powder for injection vial (167061)". Therapeutic Goods Administration (TGA). 27 May 2022. Retrieved 28 April 2023.
- ^ "Fosaprepitant MSN (Accelagen Pty Ltd)". Therapeutic Goods Administration (TGA). 11 November 2022. Archived from the original on 18 March 2023. Retrieved 28 April 2023.
- ^ "Emend- fosaprepitant dimeglumine injection, powder, lyophilized, for solution". DailyMed. 2 May 2022. Archived from the original on 1 December 2021. Retrieved 27 September 2022.
- ^ a b "Ivemend EPAR". European Medicines Agency. 17 September 2018. Archived from the original on 31 December 2021. Retrieved 27 September 2022.
- ^ Garnock-Jones KP (September 2016). "Fosaprepitant Dimeglumine: A Review in the Prevention of Nausea and Vomiting Associated with Chemotherapy". Drugs. 76 (14): 1365–72. doi:10.1007/s40265-016-0627-7. PMID 27510503. S2CID 30018182.
- ^ "Drugs.com, FDA Approves Emend (fosaprepitant dimeglumine) for Injection, Merck's New Intravenous Therapy, for Use in Combination with Other Antiemetics for Prevention of Nausea and Vomiting Caused by Chemotherapy". Archived from the original on 2008-04-09. Retrieved 2008-03-15.