 | |
| Clinical data | |
|---|---|
| Trade names | Ztalmy |
| Other names | GNX; CCD-1042; 3β-Methyl-5α-pregnan-3α-ol-20-one; 3α-Hydroxy-3β-methyl-5α-pregnan-20-one |
| License data |
|
| Routes of administration | By mouth |
| Drug class | Neurosteroid |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.210.937 |
| Chemical and physical data | |
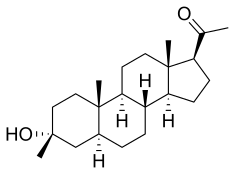
| Formula | C22H36O2 |
| Molar mass | 332.528 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Ganaxolone, sold under the brand name Ztalmy, is a medication used to treat seizures in people with cyclin-dependent kinase-like 5 (CDKL5) deficiency disorder.[1][3] Ganaxolone is a neuroactive steroid gamma-aminobutyric acid (GABA) A receptor positive modulator.[1]
The most common side effects of treatment with ganaxolone include somnolence (sleepiness), fever, excessive saliva or drooling, and seasonal allergy.[4]
Ganaxolone was approved for medical use in the United States in March 2022,[1][4][5] and in the European Union in July 2023.[2] The US Food and Drug Administration (FDA) considers it to be a first-in-class medication.[6][7]
- ^ a b c d "Ztalmy- ganaxolone suspension". DailyMed. 15 November 2022. Archived from the original on 21 January 2023. Retrieved 21 January 2023.
- ^ a b "Ztalmy EPAR". European Medicines Agency. 31 July 2023. Archived from the original on 25 August 2023. Retrieved 25 August 2023.
- ^ Carter RB, Wood PL, Wieland S, Hawkinson JE, Belelli D, Lambert JJ, et al. (March 1997). "Characterization of the anticonvulsant properties of ganaxolone (CCD 1042; 3alpha-hydroxy-3beta-methyl-5alpha-pregnan-20-one), a selective, high-affinity, steroid modulator of the gamma-aminobutyric acid(A) receptor". The Journal of Pharmacology and Experimental Therapeutics. 280 (3): 1284–1295. PMID 9067315.
- ^ a b Cite error: The named reference
Ztalmy FDA snapshotwas invoked but never defined (see the help page). - ^ Lamb YN (June 2022). "Ganaxolone: First Approval". Drugs. 82 (8): 933–940. doi:10.1007/s40265-022-01724-0. PMID 35596878.
- ^ "Advancing Health Through Innovation: New Drug Therapy Approvals 2022". U.S. Food and Drug Administration (FDA). 10 January 2023. Archived from the original on 21 January 2023. Retrieved 22 January 2023.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ New Drug Therapy Approvals 2022 (PDF). U.S. Food and Drug Administration (FDA) (Report). January 2024. Archived from the original on 14 January 2024. Retrieved 14 January 2024.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.