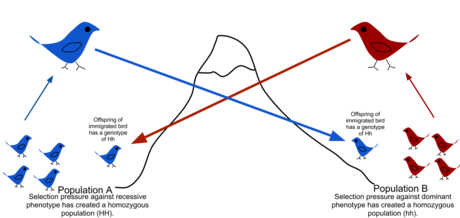
In population genetics, gene flow (also known as migration and allele flow) is the transfer of genetic material from one population to another. If the rate of gene flow is high enough, then two populations will have equivalent allele frequencies and therefore can be considered a single effective population. It has been shown that it takes only "one migrant per generation" to prevent populations from diverging due to drift.[1] Populations can diverge due to selection even when they are exchanging alleles, if the selection pressure is strong enough.[2][3] Gene flow is an important mechanism for transferring genetic diversity among populations. Migrants change the distribution of genetic diversity among populations, by modifying allele frequencies (the proportion of members carrying a particular variant of a gene). High rates of gene flow can reduce the genetic differentiation between the two groups, increasing homogeneity.[4] For this reason, gene flow has been thought to constrain speciation and prevent range expansion by combining the gene pools of the groups, thus preventing the development of differences in genetic variation that would have led to differentiation and adaptation.[5] In some cases dispersal resulting in gene flow may also result in the addition of novel genetic variants under positive selection to the gene pool of a species or population (adaptive introgression.[6])
There are a number of factors that affect the rate of gene flow between different populations. Gene flow is expected to be lower in species that have low dispersal or mobility, that occur in fragmented habitats, where there is long distances between populations, and when there are small population sizes.[7][8] Mobility plays an important role in dispersal rate, as highly mobile individuals tend to have greater movement prospects.[9] Although animals are thought to be more mobile than plants, pollen and seeds may be carried great distances by animals, water or wind. When gene flow is impeded, there can be an increase in inbreeding, measured by the inbreeding coefficient (F) within a population. For example, many island populations have low rates of gene flow due to geographic isolation and small population sizes. The Black Footed Rock Wallaby has several inbred populations that live on various islands off the coast of Australia. The population is so strongly isolated that lack of gene flow has led to high rates of inbreeding.[10]
- ^ Frankham R, Briscoe DA, Ballou JD (2002-03-14). Introduction to Conservation Genetics. Cambridge University Press. ISBN 9780521639859.
- ^ Stankowski S (May 2013). "Ecological speciation in an island snail: evidence for the parallel evolution of a novel ecotype and maintenance by ecologically dependent postzygotic isolation". Molecular Ecology. 22 (10): 2726–41. doi:10.1111/mec.12287. PMID 23506623. S2CID 39592922.
- ^ Gemmell MR, Trewick SA, Crampton JS, Vaux F, Hills SF, Daly EE, Marshall BA, Beu AG, Morgan-Richards M (2018-11-26). "Genetic structure and shell shape variation within a rocky shore whelk suggest both diverging and constraining selection with gene flow". Biological Journal of the Linnean Society. 125 (4): 827–843. doi:10.1093/biolinnean/bly142. ISSN 0024-4066.
- ^ Cite error: The named reference
:1was invoked but never defined (see the help page). - ^ Bolnick DI, Nosil P (September 2007). "Natural selection in populations subject to a migration load". Evolution; International Journal of Organic Evolution. 61 (9): 2229–43. doi:10.1111/j.1558-5646.2007.00179.x. PMID 17767592. S2CID 25685919.
- ^ Song Y, Endepols S, Klemann N, Richter D, Matuschka FR, Shih CH, et al. (August 2011). "Adaptive introgression of anticoagulant rodent poison resistance by hybridization between old world mice". Current Biology. 21 (15): 1296–301. doi:10.1016/j.cub.2011.06.043. PMC 3152605. PMID 21782438.
- ^ Hastings A, Harrison S (November 1994). "Metapopulation Dynamics and Genetics". Annual Review of Ecology and Systematics. 25 (1): 167–188. doi:10.1146/annurev.es.25.110194.001123.
- ^ "Effects of life history traits on genetic diversity in plant species". Philosophical Transactions of the Royal Society of London. Series B: Biological Sciences. 351 (1345): 1291–1298. January 1997. doi:10.1098/rstb.1996.0112.
- ^ Cunningham, Charles; Parra, Jorge E; Coals, Lucy; Beltrán, Marcela; Zefania, Sama; Székely, Tamás (2018). "Social interactions predict genetic diversification: an experimental manipulation in shorebirds". Behavioral Ecology. 29 (3): 609–618. doi:10.1093/beheco/ary012. ISSN 1045-2249. PMC 5946871. PMID 29769794.
- ^ Eldridge MD, King JM, Loupis AK, Spencer PB, Taylor AC, Pope LC, Hall GP (June 1999). "Unprecedented Low Levels of Genetic Variation and Inbreeding Depression in an Island Population of the Black-Footed Rock-Wallaby". Conservation Biology. 13 (3): 531–541. doi:10.1046/j.1523-1739.1999.98115.x. S2CID 85948161.