| IUD with progestogen | |
|---|---|
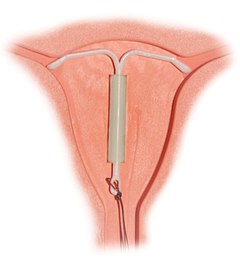 Correctly inserted IUD | |
| Background | |
| Type | Intrauterine |
| First use | 1990 (Mirena—currently available) 1976 (Progestasert—discontinued in 2001) |
| Synonyms | intrauterine system (IUS), levonorgestrel intrauterine system |
| Trade names | Mirena, Skyla, Liletta, others |
| AHFS/Drugs.com | Professional Drug Facts |
| Failure rates (first year) | |
| Perfect use | 0.1–0.2%[1] |
| Typical use | 0.1–0.2%[1] |
| Usage | |
| Duration effect | 3–8 years |
| Reversibility | 2–6 months |
| User reminders | Check thread position monthly |
| Clinic review | One month after insertion, then annually |
| Advantages and disadvantages | |
| STI protection | No |
| Periods | Menstrual irregularity, periods usually lighter or none at all |
| Weight | Potential side effect |
| Benefits | No need to remember to take daily action |
| Risks | benign ovarian cysts, transient risk of PID, uterine perforation (rare) |
A hormonal intrauterine device (IUD), also known as an intrauterine system (IUS) with progestogen and sold under the brand name Mirena among others, is an intrauterine device that releases a progestogenic hormonal agent such as levonorgestrel into the uterus.[2] It is used for birth control, heavy menstrual periods, and to prevent excessive build of the lining of the uterus in those on estrogen replacement therapy.[2] It is one of the most effective forms of birth control with a one-year failure rate around 0.2%.[1] The device is placed in the uterus and lasts three to eight years.[3][4] Fertility often returns quickly following removal.[2]
Side effects include irregular periods, benign ovarian cysts, pelvic pain, and depression.[2] Rarely uterine perforation may occur.[2] Use is not recommended during pregnancy but is safe with breastfeeding.[2] The IUD with progestogen is a type of long-acting reversible birth control.[5] It works by thickening the mucus at the opening of the cervix, stopping the buildup of the lining of the uterus, and occasionally preventing ovulation.[2]
The IUD with levonorgestrel was first approved for medical use in 1990 in Finland and in the United States in 2000.[6] It is on the World Health Organization's List of Essential Medicines.[7][8]
- ^ a b c Trussell J (2011). "Contraceptive efficacy". In Hatcher RA, Trussell J, Nelson AL, Cates Jr W, Kowal D, Policar MS (eds.). Contraceptive technology (20th revised ed.). New York: Ardent Media. pp. 779–863. ISBN 978-1-59708-004-0. ISSN 0091-9721. OCLC 781956734. Table 26–1 = "Table 3–2 Percentage of women experiencing an unintended pregnancy during the first year of typical use and the first year of perfect use of contraception, and the percentage continuing use at the end of the first year. United States" (PDF). Archived from the original (PDF) on 15 February 2017.
- ^ a b c d e f g British National Formulary: BNF 69 (69th ed.). British Medical Association. 2015. p. 556. ISBN 978-0-85711-156-2.
- ^ "Levonorgestrel intrauterine system medical facts from Drugs.com". drugs.com. Archived from the original on 1 January 2017. Retrieved 1 January 2017.
- ^ "Hormonal IUDs". www.plannedparenthood.org. Archived from the original on 24 April 2019. Retrieved 20 April 2019.
- ^ Wipf J (2015). Women's Health, An Issue of Medical Clinics of North America. Elsevier Health Sciences. p. 507. ISBN 978-0-323-37608-2. Archived from the original on 10 January 2023. Retrieved 1 September 2017.
- ^ Bradley LD, Falcone T (2008). Hysteroscopy: Office Evaluation and Management of the Uterine Cavity. Elsevier Health Sciences. p. 171. ISBN 978-0-323-04101-0. Archived from the original on 12 January 2023. Retrieved 1 September 2017.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ World Health Organization (2021). World Health Organization model list of essential medicines: 22nd list (2021). Geneva: World Health Organization. hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.