This article includes a list of references, related reading, or external links, but its sources remain unclear because it lacks inline citations. (November 2017) |
This section needs additional citations for verification. (October 2019) |

White: leading electrolyte; gray: terminating electrolyte; hatched: the analytes
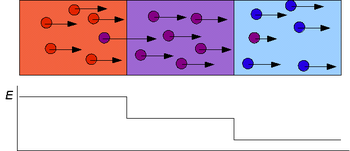
Isotachophoresis (ITP) is a technique in analytical chemistry used for selective separation and concentration of ionic analytes. It is a form of electrophoresis; charged analytes are separated based on ionic mobility, a quantity which tells how fast an ion migrates through an electric field.