| |

| |
| Names | |
|---|---|
| Other names
Mercuric thiocyanate
Mercuric sulfocyanate | |
| Identifiers | |
3D model (JSmol)
|
|
| ECHA InfoCard | 100.008.886 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
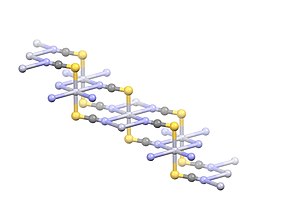
| Hg(SCN)2 | |
| Molar mass | 316.755 g/mol |
| Appearance | White monoclinic powder |
| Odor | odorless |
| Density | 3.71 g/cm3, solid |
| Melting point | 165 °C (329 °F; 438 K) (decomposes) |
| 0.069 g/100 mL | |
| Solubility | Soluble in dilute hydrochloric acid, KCN, ammonia slightly soluble in alcohol, ether |
| −96.5·10−6 cm3/mol | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
highly toxic |
| GHS labelling:[1] | |
  
| |
| Danger | |
| H300, H310, H330, H373, H410 | |
| P260, P262, P270, P271, P273, P280, P284, P301+P316, P302+P352, P304+P340, P316, P319, P320, P321, P330, P361, P364, P391, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
46 mg/kg (rat, oral) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Mercury(II) thiocyanate (Hg(SCN)2) is an inorganic chemical compound, the coordination complex of Hg2+ and the thiocyanate anion. It is a white powder. It will produce a large, winding "snake" when ignited, an effect known as the Pharaoh's serpent.[2]
- ^ "Mercuric thiocyanate (Compound)". pubchem.ncbi.nlm.nih.gov. Retrieved 31 May 2023.
- ^ Davis, T. L. (1940). "Pyrotechnic Snakes". Journal of Chemical Education. 17 (6): 268–270. doi:10.1021/ed017p268.
