 | |
| Clinical data | |
|---|---|
| Trade names | Androstalone, Ermalone, others |
| Other names | RU-143; Methylandrostanolone; Methyldihydrotestosterone; Methyl-DHT; 17α-Methyl-4,5α-dihydrotestosterone; 17α-Methyl-DHT; 17α-Methyl-5α-androstan-17β-ol-3-one; |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | By mouth |
| Drug class | Androgen; Anabolic steroid |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | Liver |
| Excretion | Urine |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.007.549 |
| Chemical and physical data | |
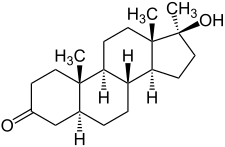
| Formula | C20H32O2 |
| Molar mass | 304.474 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Mestanolone, also known as methylandrostanolone and sold under the brand names Androstalone and Ermalone among others, is an androgen and anabolic steroid (AAS) medication which is mostly no longer used.[1][2][3][4] It is still available for use in Japan however.[2][3] It is taken by mouth.[4]
Side effects of mestanolone include symptoms of masculinization like acne, increased hair growth, voice changes, and increased sexual desire.[4] It can also cause liver damage.[4] The drug is a synthetic androgen and anabolic steroid and hence is an agonist of the androgen receptor (AR), the biological target of androgens like testosterone and dihydrotestosterone (DHT).[4][5] It has strong androgenic effects and weak anabolic effects, which make it useful for producing masculine psychological and behavioral effects.[4] The drug has no estrogenic effects.[4]
Mestanolone was discovered in 1935 and was introduced for medical use in the 1950s.[6][7][8][4] In addition to its medical use, mestanolone has been used to improve physique and performance.[4] It was used in East Germany in Olympic athletes as part of a state-sponsored doping program in the 1970s and 1980s.[4] The drug is a controlled substance in many countries and so non-medical use is generally illicit.[4]
- ^ Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 775–. ISBN 978-1-4757-2085-3.
- ^ a b "Mestanolone". Index Nominum 2000: International Drug Directory. Taylor & Francis. 2000. pp. 655–. ISBN 978-3-88763-075-1.
- ^ a b Cite error: The named reference
Drugs.comwas invoked but never defined (see the help page). - ^ a b c d e f g h i j k Llewellyn W (2009). Anabolics. Molecular Nutrition Llc. p. 241. ISBN 978-0967930473.
- ^ Kicman AT (June 2008). "Pharmacology of anabolic steroids". British Journal of Pharmacology. 154 (3): 502–521. doi:10.1038/bjp.2008.165. PMC 2439524. PMID 18500378.
- ^ Cite error: The named reference
pmid8674183was invoked but never defined (see the help page). - ^ Cite error: The named reference
RuzickaGoldberg1935was invoked but never defined (see the help page). - ^ Cite error: The named reference
pmid14086172was invoked but never defined (see the help page).