
Metabolic myopathies are myopathies that result from defects in biochemical metabolism that primarily affect muscle. They are generally genetic defects (inborn errors of metabolism) that interfere with the ability to create energy, causing a low ATP reservoir within the muscle cell.[1][2]
At the cellular level, metabolic myopathies lack some kind of enzyme or transport protein that prevents the chemical reactions necessary to create adenosine triphosphate (ATP).[1][3] ATP is often referred to as the "molecular unit of currency" of intracellular energy transfer. The lack of ATP prevents the muscle cells from being able to function properly. Some people with a metabolic myopathy never develop symptoms due to the body's ability to produce enough ATP through alternative pathways (e.g. the majority of those with AMP-deaminase deficiency are asymptomatic[1][4]).
H2O + ATP → H+ + ADP + Pi + energy → muscle contraction[5]

ATP is needed for muscle contraction by two processes:
- Firstly, ATP is needed for transport proteins to actively transport calcium ions into the sarcoplasmic reticulum (SR) of the muscle cell between muscle contractions. Afterwards, when a nerve signal is received, calcium channels in the SR open briefly and calcium rushes into the cytosol by selective diffusion (which does not use ATP) in what is called a "calcium spark." The diffusion of calcium ions into the cytosol causes the myosin strands of the myofibril to become exposed, and the myosin strands pull the actin microfilaments together. The muscle begins to contract.[6]
- Secondly, ATP is needed to allow the myosin to release and pull again, so that the muscle can contract further in what is known as the sliding filament model.[6]
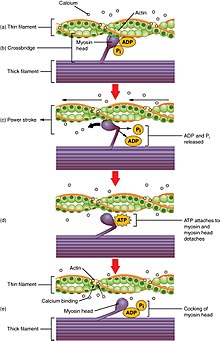
ATP is consumed at a high rate by contracting muscles. The need for ATP in muscle cells is illustrated by the phenomenon of Rigor mortis, which is the muscle rigidity that occurs in dead bodies for a short time after death. In these muscles, all the ATP has been used up and in the absence of further ATP being generated, the calcium transport proteins stop pumping calcium ions into the sarcoplasmic reticulum and the calcium ions gradually leak out. This causes the myosin proteins to grab the actin and pull once, but without further supply of ATP, cannot release and pull again. The muscles therefore remain rigid in the position at death until the binding of myosin to actin begins to break down and they become loose again.[6]
- ^ a b c Urtizberea, Jon Andoni; Severa, Gianmarco; Malfatti, Edoardo (May 2023). "Metabolic Myopathies in the Era of Next-Generation Sequencing". Genes. 14 (5): 954. doi:10.3390/genes14050954. ISSN 2073-4425. PMC 10217901. PMID 37239314.
- ^ Tobon, Alejandro (December 2013). "Metabolic myopathies". Continuum (Minneapolis, Minn.). 19 (6 Muscle Disease): 1571–97. doi:10.1212/01.CON.0000440660.41675.06. ISSN 1538-6899. PMC 10563931. PMID 24305448.
- ^ "Metabolic Myopathies". www.rheumatology.org. Retrieved 2019-11-19.
- ^ Kiani AK, Amato B, Maitz S, Nodari S, Benedetti S, Agostini F, et al. (November 2020). "Genetic test for Mendelian fatigue and muscle weakness syndromes". Acta Bio-Medica. 91 (13–S): e2020001. doi:10.23750/abm.v91i13-S.10642. PMC 8023128. PMID 33170160.
- ^ Baker JS, McCormick MC, Robergs RA (2010). "Interaction among Skeletal Muscle Metabolic Energy Systems during Intense Exercise". Journal of Nutrition and Metabolism. 2010: 905612. doi:10.1155/2010/905612. PMC 3005844. PMID 21188163.
- ^ a b c "lecture17, Energy, Use of ATP by Muscle Cells, Skeletal Muscle". www.uwyo.edu. February 18, 2005. Retrieved 2022-12-22.