 | |
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral, insufflation, intravenous, rectal, vaporization |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | Hepatic |
| Excretion | Primarily urine (renal) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
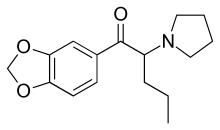
| Formula | C16H21NO3 |
| Molar mass | 275.348 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
| |
| |
| | |
Methylenedioxypyrovalerone (abbreviated MDPV, and also called monkey dust[3]) is a stimulant of the cathinone class that acts as a norepinephrine–dopamine reuptake inhibitor (NDRI).[4][5] It was first developed in the 1960s by a team at Boehringer Ingelheim.[6] Its activity at the dopamine transporter is six times stronger than at the norepinephrine transporter and it is virtually inactive at the serotonin transporter.[4] MDPV remained an obscure stimulant until around 2004 when it was reportedly sold as a designer drug. In the US, products containing MDPV and labeled as bath salts were sold as recreational drugs in gas stations, similar to the marketing for Spice and K2 as incense, until it was banned in 2011.[7]
- ^ "Substance Details 3,4-Methylenedioxypyrovalerone". Retrieved January 22, 2024.
- ^ Cite error: The named reference
EMCDDAwas invoked but never defined (see the help page). - ^ "Monkey dust "epidemic" causing drug users to experience violent hallucinations". Newsweek. August 10, 2018. Retrieved August 17, 2018.
- ^ a b Baumann MH, Partilla JS, Lehner KR, Thorndike EB, Hoffman AF, Holy M, et al. (March 2013). "Powerful cocaine-like actions of 3,4-methylenedioxypyrovalerone (MDPV), a principal constituent of psychoactive 'bath salts' products". Neuropsychopharmacology. 38 (4): 552–62. doi:10.1038/npp.2012.204. PMC 3572453. PMID 23072836.
- ^ Simmler LD, Buser TA, Donzelli M, Schramm Y, Dieu LH, Huwyler J, et al. (January 2013). "Pharmacological characterization of designer cathinones in vitro". British Journal of Pharmacology. 168 (2): 458–70. doi:10.1111/j.1476-5381.2012.02145.x. PMC 3572571. PMID 22897747.
- ^ US 3478050, Koppe H, Ludwig G, Zeile K, "1-(3',4'-methylenedioxy-phenyl)-2-pyrrolidino-alkanones-(1)", issued November 1969, assigned to CH Boehringer Sohn AG and Co and KG Boehringer Ingelheim GmbH.
- ^ Slomski A (December 2012). "A trip on "bath salts" is cheaper than meth or cocaine but much more dangerous". JAMA. 308 (23): 2445–7. doi:10.1001/jama.2012.34423. PMID 23288310.