 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Eloxatin |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a607035 |
| License data |
|
| Routes of administration | Intravenous |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Complete |
| Elimination half-life | ~10 – 25 minutes[4] |
| Excretion | Kidney |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.150.118 |
| Chemical and physical data | |
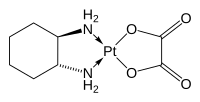
| Formula | C8H14N2O4Pt |
| Molar mass | 397.294 g·mol−1 |
| 3D model (JSmol) | |
| |
| | |
Oxaliplatin, sold under the brand name Eloxatin among others, is a cancer medication (platinum-based antineoplastic class) used to treat colorectal cancer.[5] It is given by injection into a vein.[5]
Common side effects include numbness, feeling tired, nausea, diarrhea, and low blood cell counts.[5][6] Other serious side effects include allergic reactions.[6][5] Use in pregnancy is known to harm the baby.[5] Oxaliplatin is in the platinum-based antineoplastic family of medications.[7] It is believed to work by blocking the duplication of DNA.[5]
Oxaliplatin was patented in 1976 in Japan and approved for medical use in 1996 in Europe.[8] It is on the 2023 World Health Organization's List of Essential Medicines.[9]
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 October 2023.
- ^ "Prescription medicines: registration of new generic medicines and biosimilar medicines, 2017". Therapeutic Goods Administration (TGA). 21 June 2022. Retrieved 30 March 2024.
- ^ "Eloxatin- oxaliplatin injection, solution, concentrate". DailyMed. 22 October 2019. Retrieved 26 May 2022.
- ^ Ehrsson H, Wallin I, Yachnin J (2002). "Pharmacokinetics of oxaliplatin in humans". Medical Oncology. 19 (4): 261–265. doi:10.1385/MO:19:4:261. PMID 12512920. S2CID 1068099.
- ^ a b c d e f "Oxaliplatin". The American Society of Health-System Pharmacists. Archived from the original on 21 December 2016. Retrieved 8 December 2016.
- ^ a b Oun R, Moussa YE, Wheate NJ (May 2018). "The side effects of platinum-based chemotherapy drugs: a review for chemists". Dalton Transactions. 47 (19): 6645–6653. doi:10.1039/c8dt00838h. PMID 29632935.
- ^ Apps MG, Choi EH, Wheate NJ (August 2015). "The state-of-play and future of platinum drugs". Endocrine-Related Cancer. 22 (4): R219–R233. doi:10.1530/ERC-15-0237. hdl:2123/24426. PMID 26113607.
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 513. ISBN 9783527607495. Archived from the original on 20 December 2016.
- ^ World Health Organization (2023). The selection and use of essential medicines 2023: web annex A: World Health Organization model list of essential medicines: 23rd list (2023). Geneva: World Health Organization. hdl:10665/371090. WHO/MHP/HPS/EML/2023.02.