| Pentapeptide repeat | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
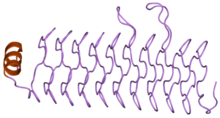 Structure of the pentapeptide repeat protein HetL.[1] | |||||||||||
| Identifiers | |||||||||||
| Symbol | Pentapeptide | ||||||||||
| Pfam | PF00805 | ||||||||||
| InterPro | IPR001646 | ||||||||||
| |||||||||||
Pentapeptide repeats are a family of sequence motifs found in multiple tandem copies in protein molecules.[2][3] Pentapeptide repeat proteins are found in all species, but they are found in many copies in cyanobacterial genomes. The repeats were first identified by Black and colleagues in the hglK protein.[4] The later Bateman et al. showed that a large family of related pentapeptide repeat proteins existed.[3] The function of these repeats is uncertain in most proteins. However, in the MfpA protein a DNA gyrase inhibitor it has been suggested that the pentapeptide repeat structure mimics the structure of DNA.[5] The repeats form a regular right handed four sided beta helix structure known as the Rfr-fold.
- ^ Ni S, Sheldrick GM, Benning MM, Kennedy MA (January 2009). "The 2 Å resolution crystal structure of HetL, a pentapeptide repeat protein involved in regulation of heterocyst differentiation in the cyanobacterium Nostoc sp. strain PCC 7120". J. Struct. Biol. 165 (1): 47–52. doi:10.1016/j.jsb.2008.09.010. PMID 18952182.
- ^ Vetting MW, Hegde SS, Fajardo JE, et al. (January 2006). "Pentapeptide repeat proteins". Biochemistry. 45 (1): 1–10. doi:10.1021/bi052130w. PMC 2566302. PMID 16388575.
- ^ a b Bateman A, Murzin AG, Teichmann SA (June 1998). "Structure and distribution of pentapeptide repeats in bacteria". Protein Sci. 7 (6): 1477–80. doi:10.1002/pro.5560070625. PMC 2144021. PMID 9655353.
- ^ Black K, Buikema WJ, Haselkorn R (November 1995). "The hglK gene is required for localization of heterocyst-specific glycolipids in the cyanobacterium Anabaena sp. strain PCC 7120". J. Bacteriol. 177 (22): 6440–8. doi:10.1128/jb.177.22.6440-6448.1995. PMC 177493. PMID 7592418.
- ^ Hegde SS, Vetting MW, Roderick SL, et al. (June 2005). "A fluoroquinolone resistance protein from Mycobacterium tuberculosis that mimics DNA". Science. 308 (5727): 1480–3. Bibcode:2005Sci...308.1480H. doi:10.1126/science.1110699. PMID 15933203. S2CID 20194294.