| |
| Names | |
|---|---|
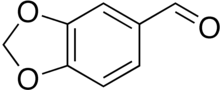
| Preferred IUPAC name
2H-1,3-Benzodioxole-5-carbaldehyde | |
| Other names
Heliotropin; Heliotropine; Piperonyl aldehyde; Protocatechuic aldehyde methylene ether; 3,4-methylenedioxybenzaldehyde;
| |
| Identifiers | |
3D model (JSmol)
|
|
| 131691 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.004.009 |
| EC Number |
|
| 4186 | |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C8H6O3 | |
| Molar mass | 150.133 g·mol−1 |
| Appearance | Colorless crystals[1] |
| Density | 1.337 g/cm3 |
| Melting point | 37 °C (99 °F; 310 K)[1] |
| Boiling point | 263 °C (505 °F; 536 K)[1] |
| Soluble in 500 parts[1] | |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H317 | |
| P261, P272, P280, P302+P352, P321, P333+P313, P363, P501 | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
2700 mg/kg (orally in rats)[1] |
| Legal status | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Piperonal, also known as heliotropin, is an organic compound which is commonly found in fragrances and flavors.[3] The molecule is structurally related to other aromatic aldehydes such as benzaldehyde and vanillin.
- ^ a b c d e Budavari, Susan, ed. (1996), The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals (12th ed.), Merck, ISBN 0911910123
- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-15.
- ^ Karl-Georg Fahlbusch, Franz-Josef Hammerschmidt, Johannes Panten, Wilhelm Pickenhagen, Dietmar Schatkowski, Kurt Bauer, Dorothea Garbe and Horst Surburg "Flavors and Fragrances" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2003. doi:10.1002/14356007.a11_141