 | |
| Clinical data | |
|---|---|
| Trade names | Trulance |
| Other names | SP-304 |
| License data |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
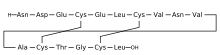
| Formula | C65H104N18O26S4 |
| Molar mass | 1681.89 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Plecanatide, sold under the brand name Trulance, is a medication for the treatment of chronic idiopathic constipation (CIC)[3] and irritable bowel syndrome with constipation.[4] It is being launched in India under the brand name "Plecasoft" (Gromaxx, Zuventus Health care ltd, India). Plecanatide is an agonist of guanylate cyclase-C. Plecanatide increases intestinal transit and fluid through a buildup of cGMP.[5][6]
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 Oct 2023.
- ^ "Summary Basis of Decision (SBD) for Trulance". Health Canada. 23 October 2014. Retrieved 29 May 2022.
- ^ "FDA approves Trulance for Chronic Idiopathic Constipation". FDA.gov. U.S. Food and Drug Administration. Retrieved 20 January 2017.
- ^ Miner, Philip B (2020-02-01). "Plecanatide for the treatment of constipation-predominant irritable bowel syndrome". Expert Review of Gastroenterology & Hepatology. 14 (2): 71–84. doi:10.1080/17474124.2020.1722101. ISSN 1747-4124. PMID 31985305. S2CID 210922857.
- ^ "TRULANCE package insert" (PDF). Trulance website. Synergy Pharmaceuticals Inc. 420 Lexington Avenue, Suite 2012 New York, New York 10170. Retrieved 20 January 2017.
- ^ Thomas RH, Luthin DR (June 2015). "Current and emerging treatments for irritable bowel syndrome with constipation and chronic idiopathic constipation: focus on prosecretory agents". Pharmacotherapy. 35 (6): 613–30. doi:10.1002/phar.1594. PMID 26016701. S2CID 24825251.