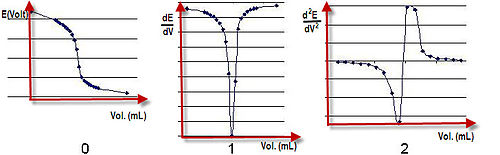
In analytical chemistry, potentiometric titration is a technique similar to direct titration of a redox reaction. It is a useful means of characterizing an acid. No indicator is used; instead the electric potential is measured across the analyte, typically an electrolyte solution. To do this, two electrodes are used, an indicator electrode (the glass electrode and metal ion indicator electrode) and a reference electrode. Reference electrodes generally used are hydrogen electrodes, calomel electrodes, and silver chloride electrodes. The indicator electrode forms an electrochemical half-cell with the interested ions in the test solution. The reference electrode forms the other half-cell.
The overall electric potential is calculated as
Esol is the potential drop over the test solution between the two electrodes. Ecell is recorded at intervals as the titrant is added. A graph of potential against volume added can be drawn and the end point of the reaction is halfway between the jump in voltage. Ecell depends on the concentration of the interested ions with which the indicator electrode is in contact. For example, the electrode reaction may be
As the concentration of Mn+ changes, the Ecell changes correspondingly. Thus the potentiometric titration involve measurement of Ecell with the addition of titrant. Types of potentiometric titration include acid–base titration (total alkalinity and total acidity), redox titration (HI/HY and cerate), precipitation titration (halides), and complexometric titration (free EDTA and Antical #5).

