| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Pyridazine[1] | |||
| Systematic IUPAC name
1,2-Diazabenzene | |||
| Other names
1,2-Diazine
Orthodiazine Oizine | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 103906 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.005.478 | ||
| EC Number |
| ||
| 49310 | |||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C4H4N2 | |||
| Molar mass | 80.090 g·mol−1 | ||
| Appearance | Colorless liquid | ||
| Density | 1.107 g/cm3 | ||
| Melting point | −8 °C (18 °F; 265 K) | ||
| Boiling point | 208 °C (406 °F; 481 K) | ||
| miscible | |||
| Solubility | miscible in dioxane, ethanol soluble in benzene, diethyl ether negligible in cyclohexane, ligroin | ||
Refractive index (nD)
|
1.52311 (23.5 °C) | ||
| Thermochemistry | |||
Std enthalpy of
formation (ΔfH⦵298) |
224.9 kJ/mol | ||
| Hazards | |||
| GHS labelling:[2] | |||

| |||
| Warning | |||
| H302, H315, H319, H335 | |||
| P261, P264, P264+P265, P270, P271, P280, P301+P317, P302+P352, P304+P340, P305+P351+P338, P319, P321, P330, P332+P317, P337+P317, P362+P364, P403+P233, P405, P501 | |||
| Flash point | 85 °C (185 °F; 358 K) | ||
| Related compounds | |||
Related compounds
|
|||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
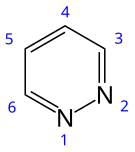
Pyridazine is an aromatic, heterocyclic, organic compound with the molecular formula C4H4N2. It contains a six-membered ring with two adjacent nitrogen atoms.[3] It is a colorless liquid with a boiling point of 208 °C. It is isomeric with two other diazine (C4H4N2) rings, pyrimidine and pyrazine.
- ^ "Front Matter". Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. p. 141. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
- ^ "Pyridazine". pubchem.ncbi.nlm.nih.gov.
- ^ Gumus, S. (2011). "A computational study on substituted diazabenzenes" (PDF). Turk J Chem. 35: 803–808. Archived from the original (PDF) on 2016-03-03. Retrieved 2014-04-10.