 | |
| Clinical data | |
|---|---|
| Trade names | Actonel, Atelvia, Benet, others |
| AHFS/Drugs.com | Monograph |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 0.63% |
| Protein binding | ~24% |
| Metabolism | None |
| Elimination half-life | 1.5 h |
| Excretion | Kidney and fecal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.116.436 |
| Chemical and physical data | |
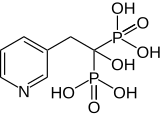
| Formula | C7H11NO7P2 |
| Molar mass | 283.113 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Risedronic acid, often used as its sodium salt risedronate sodium, is a bisphosphonate.[1] It slows down the cells which break down bone.[1] It's used to treat or prevent osteoporosis, and treat Paget's disease of bone.[1] It is taken by mouth.[1]
It was patented in 1984 and approved for medical use in 1998.[2]
- ^ a b c d e "Actonel- risedronate sodium tablet, film coated". DailyMed. 1 November 2019. Retrieved 28 June 2022.
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 523. ISBN 9783527607495.