| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
4-Methylbenzene-1-sulfonyl chloride | |
| Other names
Tosyl chloride, p-toluenesulfonyl chloride, p-TsCl, TsCl
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.002.441 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C7H7ClO2S | |
| Molar mass | 190.65 g/mol |
| Appearance | White solid |
| Melting point | 65 to 69 °C (149 to 156 °F; 338 to 342 K) |
| Boiling point | 134 °C (273 °F; 407 K) at 10 mmHg |
| Reacts with water | |
| Hazards[1] | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Releases HCl on contact with water |
| NFPA 704 (fire diamond) | |
| Flash point | 128 °C (262 °F; 401 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
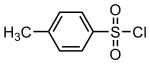
4-Toluenesulfonyl chloride (p-toluenesulfonyl chloride, toluene-p-sulfonyl chloride) is an organic compound with the formula CH3C6H4SO2Cl. This white, malodorous solid is a reagent widely used in organic synthesis.[2] Abbreviated TsCl or TosCl, it is a derivative of toluene and contains a sulfonyl chloride (−SO2Cl) functional group.
- ^ "P-TOLUENESULFONYL CHLORIDE (TOSYL CHLORIDE)".
- ^ Whitaker, D. T.; Whitaker, K. S.; Johnson, C. R.; Haas, J. (2006). "P-Toluenesulfonyl Chloride". p-Toluenesulfonyl Chloride. Encyclopedia of Reagents for Organic Synthesis. New York: John Wiley. doi:10.1002/047084289X.rt136.pub2. ISBN 978-0471936237. Archived from the original on 2016-03-05. Retrieved 2013-05-28.
