 | |
| Clinical data | |
|---|---|
| Trade names | Midamor, others |
| Other names | MK-870 |
| AHFS/Drugs.com | Monograph |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Readily absorbed, 15–25% |
| Protein binding | ~23% |
| Metabolism | Nil |
| Onset of action | 2 hours (peak at 6–10 hours, duration ~24 hours) |
| Elimination half-life | 6 to 9 hours |
| Excretion | urine (20–50%), feces (40%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.018.205 |
| Chemical and physical data | |
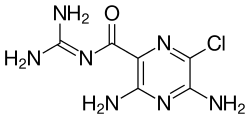
| Formula | C6H8ClN7O |
| Molar mass | 229.63 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 240.5 to 241.5 °C (464.9 to 466.7 °F) |
| |
| |
| (verify) | |
Amiloride, sold under the trade name Midamor among others, is a medication typically used with other medications to treat high blood pressure or swelling due to heart failure or cirrhosis of the liver.[1][2] Amiloride is classified as a potassium-sparing diuretic. Amiloride is often used together with another diuretic, such as a thiazide or loop diuretic.[2] It is taken by mouth.[1] Onset of action is about two hours and it lasts for about a day.[2]
Common side effects include high blood potassium, vomiting, loss of appetite, rash, and headache.[1] The risk of high blood potassium is greater in those with kidney problems, diabetes, and those who are older.[1] Amiloride blocks the epithelial sodium channel (ENaC) in the late distal tubule, connecting tubule, and collecting duct of the nephron,[3] which both reduces absorption of sodium ion from the lumen of the nephron and reduces excretion of potassium ion into the lumen.[2]
Amiloride was developed in 1967.[4] It is on the World Health Organization's List of Essential Medicines.[5]
- ^ a b c d "Amiloride Hydrochloride". The American Society of Health-System Pharmacists. Archived from the original on 27 December 2016. Retrieved 8 December 2016.
- ^ a b c d World Health Organization (2009). Stuart MC, Kouimtzi M, Hill SR (eds.). WHO Model Formulary 2008. World Health Organization. pp. 328, 330. hdl:10665/44053. ISBN 9789241547659.
- ^ Nesterov V, Dahlmann A, Krueger B, Bertog M, Loffing J, Korbmacher C (November 2012). "Aldosterone-dependent and -independent regulation of the epithelial sodium channel (ENaC) in mouse distal nephron". American Journal of Physiology. Renal Physiology. 303 (9): F1289–F1299. doi:10.1152/ajprenal.00247.2012. PMID 22933298.
- ^ Progress in Drug Research/Fortschritte der Arzneimittelforschung/Progrés des recherches pharmaceutiques. Birkhäuser. 2013. p. 210. ISBN 9783034870948. Archived from the original on 2016-12-28.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.