 | |
 | |
| Clinical data | |
|---|---|
| Other names | PCN-101; HR-071603 |
| Addiction liability | Moderate |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
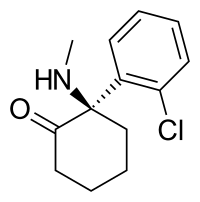
| Formula | C13H16ClNO |
| Molar mass | 237.73 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Arketamine (developmental code names PCN-101, HR-071603), also known as (R)-ketamine or (R)-(−)-ketamine, is the (R)-(−) enantiomer of ketamine.[1][2][3] Similarly to racemic ketamine and esketamine, the S(+) enantiomer of ketamine, arketamine is biologically active; however, it is less potent as an NMDA receptor antagonist and anesthetic and thus has never been approved or marketed for clinical use as an enantiopure drug.[1][3] Arketamine is currently in clinical development as a novel antidepressant.[4][5]
Relative to esketamine, arketamine possesses 4 to 5 times lower affinity for the PCP site of the NMDA receptor.[2][6] In accordance, arketamine is significantly less potent than racemic ketamine and especially esketamine in terms of anesthetic, analgesic, and sedative-hypnotic effects.[6] Racemic ketamine has weak affinity for the sigma receptor, where it acts as an agonist, whereas esketamine binds negligibly to this receptor, and so the sigma receptor activity of racemic ketamine lies in arketamine.[7] It was suggested that this action of arketamine may play a role in the hallucinogenic effects of racemic ketamine and that it may be responsible for the lowering of the seizure threshold seen with racemic ketamine.[7] However several subsequent studies have indicated that esketamine is more likely to induce dissociative events,[8][9] while studies in patients undergoing electroconvulsive therapy suggested that esketamine is a potent inducer of seizures.[10] Esketamine inhibits the dopamine transporter about 8-fold more potently than does arketamine, and so is about 8 times more potent as a dopamine reuptake inhibitor.[11] Arketamine and esketamine possess similar potency for interaction with the muscarinic acetylcholine receptors.[12]
- ^ a b Ganellin CR, Triggle DJ (21 November 1996). Dictionary of Pharmacological Agents. CRC Press. pp. 1188–. ISBN 978-0-412-46630-4.
- ^ a b Yew DT (6 March 2015). Ketamine: Use and Abuse. Taylor & Francis. pp. 269–. ISBN 978-1-4665-8340-5.
- ^ a b Singh JB, Fedgchin M, Daly E, Xi L, Melman C, De Bruecker G, et al. (September 2016). "Intravenous Esketamine in Adult Treatment-Resistant Depression: A Double-Blind, Double-Randomization, Placebo-Controlled Study". Biological Psychiatry. 80 (6): 424–431. doi:10.1016/j.biopsych.2015.10.018. PMID 26707087.
- ^ Cite error: The named reference
Hashimoto2019was invoked but never defined (see the help page). - ^ Cite error: The named reference
AdisInsight-HR-071603was invoked but never defined (see the help page). - ^ a b Barash P, Cullen BF, Stoelting RK, Cahalan M, Stock MC, Ortega R (28 March 2012). Clinical Anesthesia. Lippincott Williams & Wilkins. pp. 456–. ISBN 978-1-4511-4795-7.
- ^ a b Verster JC, Brady K, Galanter M, Conrod P, eds. (6 July 2012). Drug Abuse and Addiction in Medical Illness: Causes, Consequences and Treatment. Springer Science & Business Media. pp. 205–. ISBN 978-1-4614-3375-0.
- ^ Vollenweider FX, Leenders KL, Oye I, Hell D, Angst J (February 1997). "Differential psychopathology and patterns of cerebral glucose utilisation produced by (S)- and (R)-ketamine in healthy volunteers using positron emission tomography (PET)". European Neuropsychopharmacology. 7 (1): 25–38. doi:10.1016/S0924-977X(96)00042-9. PMID 9088882. S2CID 26861697.
- ^ Engelhardt W (March 1997). "[Recovery and psychomimetic reactions following S-(+)-ketamine]". Der Anaesthesist. 46 (Suppl 1): S38–S42. doi:10.1007/pl00002463. PMID 9163277. S2CID 24966884.
- ^ Zavorotnyy M, Kluge I, Ahrens K, Wohltmann T, Köhnlein B, Dietsche P, et al. (December 2017). "S -ketamine compared to etomidate during electroconvulsive therapy in major depression". European Archives of Psychiatry and Clinical Neuroscience. 267 (8): 803–813. doi:10.1007/s00406-017-0800-3. PMID 28424861. S2CID 22725552.
- ^ Nishimura M, Sato K (October 1999). "Ketamine stereoselectively inhibits rat dopamine transporter". Neuroscience Letters. 274 (2): 131–134. doi:10.1016/s0304-3940(99)00688-6. PMID 10553955. S2CID 10307361.
- ^ Vuyk J, Schraag S (6 December 2012). Advances in Modelling and Clinical Application of Intravenous Anaesthesia. Springer Science & Business Media. pp. 270–. ISBN 978-1-4419-9192-8.