 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Nuvigil, others |
| Other names | (R)-Modafinil; R-Modafinil; (R)-(–)-Modafinil; (–)-Modafinil; CRL-40982; CEP-10952 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a602016 |
| Pregnancy category |
|
| Dependence liability | Low |
| Routes of administration | Oral (tablets)[1] |
| Drug class | Atypical dopamine reuptake inhibitor; wakefulness-promoting agent |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Unknown (due to poor aqueous solubility;[1] but modafinil is 40–65% based on urinary excretion)[4][5] |
| Protein binding | Unknown (but for modafinil is moderate, primarily to albumin)[6][7][1] |
| Metabolism | Liver, including CYP3A4 and other enzymes (hydrolytic amidation, sulfoxidation, aromatic ring hydroxylation, and glucuronide conjugation)[6][8][7] |
| Metabolites | • Armodafinil acid[6][7] • Modafinil sulfone[6][7] |
| Onset of action | 1.5–6.5 h (range 0.5–11 h) (peak)[7][8] |
| Elimination half-life | 10–17 hours[6][7][5] |
| Duration of action | Up to 13.5 hours[9] |
| Excretion | Unknown (but modafinil is excreted 80% in urine and 1.0% in feces)[1] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.207.833 |
| Chemical and physical data | |
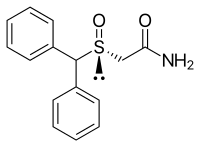
| Formula | C15H15NO2S |
| Molar mass | 273.35 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Armodafinil, sold under the brand name Nuvigil, is a wakefulness-promoting medication which is used to treat excessive daytime sleepiness associated with obstructive sleep apnea, narcolepsy, and shift work disorder.[1] It is also used off-label for certain other indications.[10] The drug is taken by mouth.[1]
Side effects of armodafinil include headache, nausea, dizziness, and insomnia.[1] Armodafinil acts as a selective atypical dopamine reuptake inhibitor (DRI) and hence as an indirect dopamine receptor agonist.[1][5][11] However, other mechanisms might also be involved in its effects.[1][5][11] Chemically, armodafinil is the enantiopure (R)-(–)-enantiomer of the racemic mixture modafinil (brand name Provigil).[1][4][5] Both enantiomers of modafinil are active as DRIs and wakefulness-promoting agents, but armodafinil is more potent and longer-acting.[4][5]
Armodafinil is produced by the pharmaceutical company Cephalon[12] and was approved by the United States Food and Drug Administration (FDA) in 2007.[13][14] In 2016, the FDA granted Mylan rights for the first generic version of armodafinil to be marketed in the United States.[15]
- ^ a b c d e f g h i j https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/021875s023lbl.pdf
- ^ "Prescription medicines: registration of new chemical entities in Australia, 2015". Therapeutic Goods Administration (TGA). 21 June 2022. Retrieved 10 April 2023.
- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-03.
- ^ a b c Sousa A, Dinis-Oliveira RJ (2020). "Pharmacokinetic and pharmacodynamic of the cognitive enhancer modafinil: Relevant clinical and forensic aspects". Subst Abus. 41 (2): 155–173. doi:10.1080/08897077.2019.1700584. PMID 31951804.
- ^ a b c d e f Hersey M, Tanda G (2024). "Modafinil, an atypical CNS stimulant?". Pharmacological Advances in Central Nervous System Stimulants. Adv Pharmacol. Vol. 99. pp. 287–326. doi:10.1016/bs.apha.2023.10.006. ISBN 978-0-443-21933-7. PMID 38467484.
- ^ a b c d e Niemegeers P, Maudens KE, Morrens M, Patteet L, Joos L, Neels H, Sabbe BG (September 2012). "Pharmacokinetic evaluation of armodafinil for the treatment of bipolar depression". Expert Opin Drug Metab Toxicol. 8 (9): 1189–1197. doi:10.1517/17425255.2012.708338. PMID 22803602.
- ^ a b c d e f Garnock-Jones KP, Dhillon S, Scott LJ (September 2009). "Armodafinil". CNS Drugs. 23 (9): 793–803. doi:10.2165/11203290-000000000-00000. PMID 19689169.
- ^ a b Lankford DA (April 2008). "Armodafinil: a new treatment for excessive sleepiness". Expert Opin Investig Drugs. 17 (4): 565–573. doi:10.1517/13543784.17.4.565. PMID 18363520.
- ^ Russo M (2009). "Pharmacotherapy of Excessive Sleepiness: Focus on Armodafinil". Clinical Medicine. Therapeutics. 1: CMT.S1994. doi:10.4137/CMT.S1994. ISSN 1179-1713.
- ^ Cite error: The named reference
repurposed2021was invoked but never defined (see the help page). - ^ "NUVIGIL (armodafinil) tablets, for oral use" (PDF). Cephalon, Inc., Teva Pharmaceutical Industries Ltd. U.S. Food and Drug Administration. Archived from the original (PDF) on 2018-01-07. Retrieved 2013-11-23.
- ^ "CDER Drug and Biologic Approvals for Calendar Year 2007" (PDF). Archived from the original (PDF) on February 3, 2014. Retrieved January 21, 2008.
- ^ "Search results from the "OB_Rx" table for query on "021875."", Orange Book, U.S. Food and Drug Administration, March 2012, retrieved April 30, 2012
- ^ "Mylan Launches First Generic of Nuvigil® Tablets". MediaRoom. Archived from the original on 2017-02-11. Retrieved February 9, 2017.