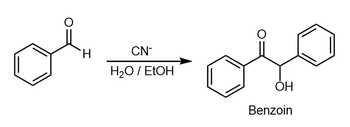
In organic chemistry, the benzoin addition is an addition reaction involving two aldehydes (−CH=O). The reaction generally occurs between aromatic aldehydes or glyoxals (OCH=CHO),[1][2] and results in formation of an acyloin (−C(O)CH(OH)−). In the classic example, benzaldehyde is converted to benzoin (PhCH(OH)C(O)Ph).[3]
The benzoin condensation was first reported in 1832 by Justus von Liebig and Friedrich Wöhler during their research on bitter almond oil.[4] The catalytic version of the reaction involving cyanide was developed by Nikolay Zinin in the late 1830s.[5][6]
- ^ Menon, Rajeev S.; Biju, Akkattu T.; Nair, Vijay (2016). "Recent advances in N-heterocyclic carbene (NHC)-catalysed benzoin reactions". Beilstein Journal of Organic Chemistry. 12: 444–461. doi:10.3762/bjoc.12.47. PMC 4901930. PMID 27340440.
- ^ Enders, Dieter; Niemeier, Oliver; Henseler, Alexander (2007). "Organocatalysis by N-Heterocyclic Carbenes". Chemical Reviews. 107 (12): 5606–5655. doi:10.1021/cr068372z. PMID 17956132.
- ^ Roger Adams; C. S. Marvel (1921). "Benzoin". Organic Syntheses. 1: 33. doi:10.15227/orgsyn.001.0033.
- ^ F. Wöhler, J. Liebig (1832). "Untersuchungen über das Radikal der Benzoesäure" [Studies on the radicals of benzoic acid]. Annalen der Pharmacie (in German). 3 (3): 249–282. doi:10.1002/jlac.18320030302. hdl:2027/hvd.hxdg3f.
- ^ N. Zinin (1839). "Beiträge zur Kenntniss einiger Verbindungen aus der Benzoylreihe" [Contributions to the knowledge of some compounds from the benzoyl series]. Annalen der Pharmacie (in German). 31 (3): 329–332. doi:10.1002/jlac.18390310312. Archived from the original on 2022-07-09. Retrieved 2020-09-11.
- ^ N. Zinin (1840). "Ueber einige Zersetzungsprodukte des Bittermandelöls" [Study of some decomposition products of bitter almond oil]. Annalen der Pharmacie (in German). 34 (2): 186–192. doi:10.1002/jlac.18400340205. Archived from the original on 2022-07-09. Retrieved 2019-06-28.