 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Pharmacokinetic data | |
| Elimination half-life | 2.5 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
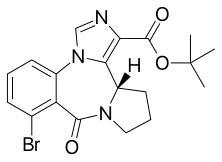
| Formula | C19H20BrN3O3 |
| Molar mass | 418.291 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Bretazenil (Ro16-6028) is an imidazopyrrolobenzodiazepine[1] anxiolytic drug which is derived from the benzodiazepine family, and was invented in 1988. It is most closely related in structure to the GABA antagonist flumazenil, although its effects are somewhat different. It is classified as a high-potency benzodiazepine due to its high affinity binding to benzodiazepine binding sites[2][3] where it acts as a partial agonist.[4] Its profile as a partial agonist and preclinical trial data suggests that it may have a reduced adverse effect profile.[5] In particular bretazenil has been proposed to cause a less strong development of tolerance and withdrawal syndrome.[6] Bretazenil differs from traditional 1,4-benzodiazepines by being a partial agonist and because it binds to α1, α2, α3, α4, α5 and α6 subunit containing GABAA receptor benzodiazepine receptor complexes. 1,4-benzodiazepines bind only to α1, α2, α3 and α5 GABAA benzodiazepine receptor complexes.[1]
- ^ a b Pym LJ, Cook SM, Rosahl T, McKernan RM, Atack JR (November 2005). "Selective labelling of diazepam-insensitive GABAA receptors in vivo using [3H]Ro 15-4513". British Journal of Pharmacology. 146 (6): 817–25. doi:10.1038/sj.bjp.0706392. PMC 1751217. PMID 16184188.
- ^ Giusti P, Guidetti G, Costa E, Guidotti A (June 1991). "The preferential antagonism of pentylenetetrazole proconflict responses differentiates a class of anxiolytic benzodiazepines with potential antipanic action". The Journal of Pharmacology and Experimental Therapeutics. 257 (3): 1062–8. PMID 1675286.
- ^ Finn DA, Gee KW (November 1993). "A comparison of Ro 16-6028 with benzodiazepine receptor 'full agonists' on GABAA receptor function". European Journal of Pharmacology. 247 (3): 233–7. doi:10.1016/0922-4106(93)90190-K. PMID 7905829.
- ^ Haefely W, Facklam M, Schoch P, Martin JR, Bonetti EP, Moreau JL, et al. (1992). "Partial agonists of benzodiazepine receptors for the treatment of epilepsy, sleep, and anxiety disorders". Advances in Biochemical Psychopharmacology. 47: 379–94. PMID 1324584.
- ^ Kunovac JL, Stahl SM (December 1995). "Future directions in anxiolytic pharmacotherapy". The Psychiatric Clinics of North America. 18 (4): 895–909. doi:10.1016/S0193-953X(18)30030-3. PMID 8748388.
- ^ Płaźnik A (1995). "Pharmacology of tolerance to benzodiazepine receptor ligands". Polish Journal of Pharmacology. 47 (6): 489–99. PMID 8868371.