| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Carbamic acid[1] | |||
| Other names
Aminomethanoic acid
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
| KEGG | |||
| MeSH | Carbamic+acid | ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| NH3CO2 | |||
| Molar mass | 61.040 g·mol−1 | ||
| Related compounds | |||
Related compounds
|
Formamide Dithiocarbamate Carbonic acid Urea Ethyl carbamate Sulfamic acid | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
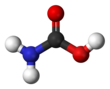
Carbamic acid, which might also be called aminoformic acid or aminocarboxylic acid,[2] is the chemical compound with the formula H2NCOOH. It can be obtained by the reaction of ammonia NH3 and carbon dioxide CO2 at very low temperatures, which also yields ammonium carbamate [NH4]+[NH2CO2]−. The compound is stable only up to about 250 K (−23 °C); at higher temperatures it decomposes into those two gases.[3] The solid apparently consists of dimers, with the two molecules connected by hydrogen bonds between the two carboxyl groups –COOH.[4]
Carbamic acid could be seen as both an amine and carboxylic acid, and therefore an amino acid;[3] however, the attachment of the carboxyl group –COOH directly to the nitrogen atom (without any intermediate carbon chain) makes it behave very differently from the amino acids with intermediate carbon chain. (Glycine NH2CH2COOH is generally considered to be the simplest amino acid.) The hydroxyl group –OH attached to the carbon also excludes it from the amide class.
The term "carbamic acid" is also used generically for any compounds of the form RR′NCOOH, where R and R′ are organic groups or hydrogen.[5]
Deprotonation of a carbamic acid yields a carbamate anion RR′NCOO−, the salts of which can be relatively stable. Carbamate is also a term used for esters of carbamic acids, such as methyl carbamate H2N−C(=O)−OCH3. The carbamoyl functional group RR′N–C(=O)– (often denoted by Cbm) is the carbamic acid molecule minus the OH part of the carboxyl.
- ^ International Union of Pure and Applied Chemistry (2014). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013. The Royal Society of Chemistry. p. 778. doi:10.1039/9781849733069. ISBN 978-0-85404-182-4.
- ^ "PubChem Compound Summary for CID 277, Carbamic acid". National Center for Biotechnology Information. 2020. Retrieved October 10, 2020.
- ^ a b R. K. Khanna and M. H. Moore (1999): "Carbamic acid: molecular structure and IR spectra". Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, volume 55, issue 5, pages 961-967. doi:10.1016/S1386-1425(98)00228-5 PMID 10347902Bibcode:1999AcSpA..55..961K
- ^ J. B. Bossa, P. Theulé, F. Duvernay, F. Borget and T. Chiavassa (2008): "Carbamic acid and carbamate formation in NH3:CO2 ices – UV irradiation versus thermal processes". Astronomy and Astrophysics, volume 492, issue 3, pages 719-724. doi:10.1051/0004-6361:200810536
- ^ Cite error: The named reference
dijk2007was invoked but never defined (see the help page).