 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ˌsɛfəˈlɛksɪn/ |
| Trade names | Keflex, others |
| Other names | cephalexin, cephalexin (BAN UK), cephalexin (USAN US) |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682733 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth |
| Drug class | First-generation cephalosporin |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Well absorbed |
| Protein binding | 15% |
| Metabolism | 80% excreted unchanged in urine within 6 hours of administration |
| Elimination half-life | 0.6–1.2 hours[3] |
| Excretion | Kidney |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.036.142 |
| Chemical and physical data | |
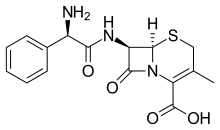
| Formula | C16H17N3O4S |
| Molar mass | 347.39 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 326.8 °C (620.2 °F) |
| |
| |
| (verify) | |
Cefalexin, also spelled cephalexin, is an antibiotic that can treat a number of bacterial infections.[4] It kills gram-positive and some gram-negative bacteria by disrupting the growth of the bacterial cell wall.[4] Cefalexin is a β-lactam antibiotic within the class of first-generation cephalosporins.[4] It works similarly to other agents within this class, including intravenous cefazolin, but can be taken by mouth.[5]
Cefalexin can treat certain bacterial infections, including those of the middle ear, bone and joint, skin, and urinary tract.[4] It may also be used for certain types of pneumonia and strep throat and to prevent bacterial endocarditis.[4] Cefalexin is not effective against infections caused by methicillin-resistant Staphylococcus aureus (MRSA), most Enterococcus, or Pseudomonas.[4] Like other antibiotics, cefalexin cannot treat viral infections, such as the flu, common cold or acute bronchitis.[4] Cefalexin can be used in those who have mild or moderate allergies to penicillin.[4] However, it is not recommended in those with severe penicillin allergies.[4]
Common side effects include stomach upset and diarrhea.[4] Allergic reactions or infections with Clostridium difficile, a cause of diarrhea, are also possible.[4] Use during pregnancy or breast feeding does not appear to be harmful to the fetus.[4][6][7] It can be used in children and those over 65 years of age.[4] Those with kidney problems may require a decrease in dose.[4]
Cefalexin was developed in 1967.[8][9][10] It was first marketed in 1969 and 1970 under the names Keflex and Ceporex, among others.[11][12] It is available as a generic medication.[4][13] It is on the World Health Organization's List of Essential Medicines.[14] In 2021, it was the 96th most commonly prescribed medication in the United States, with more than 7 million prescriptions.[15][16] In Canada, it was the fifth most common antibiotic used in 2013.[17] In Australia, it is one of the top 15 most prescribed medications.[18]
- ^ "Cephalexin Use During Pregnancy". Drugs.com. 28 December 2018. Archived from the original on 7 February 2020. Retrieved 7 February 2020.
- ^ Cite error: The named reference
Lexylan PIwas invoked but never defined (see the help page). - ^ McEvoy, G.K. (ed.). American Hospital Formulary Service — Drug Information 95. Bethesda, MD: American Society of Hospital Pharmacists, Inc., 1995 (Plus Supplements 1995)., p. 166
- ^ a b c d e f g h i j k l m n o "Cephalexin". The American Society of Health-System Pharmacists. Archived from the original on 1 May 2014. Retrieved 21 April 2014.
- ^ Brunton LL (2011). "Chapter 53: Penicillins, Cephalosporins, and Other β-Lactam Antibiotics". Goodman & Gilman's pharmacological basis of therapeutics (12th ed.). New York: McGraw-Hill. ISBN 978-0071624428.
- ^ "Prescribing medicines in pregnancy database". Australian Government. 3 March 2014. Archived from the original on 8 April 2014. Retrieved 22 April 2014.
- ^ Jones W (2013). Breastfeeding and Medication. Routledge. p. 227. ISBN 9781136178153. Archived from the original on 8 September 2017.
- ^ Hey E, ed. (2007). Neonatal formulary 5 drug use in pregnancy and the first year of life (5th ed.). Blackwell. p. 67. ISBN 9780470750353. Archived from the original on 8 September 2017.>
- ^ US patent 3275626, Morin RB, Jackson BG, "Penicillin conversion via sulfoxide", published 1966-09-27, issued 1966-09-27, assigned to Eli Lilly and Co "Espacenet - Bibliographic data". Archived from the original on 25 September 2022. Retrieved 3 March 2020.
- ^ US patent 3507861, Morin RB, Jackson BG, "Certain 3-methyl-cephalosporin compounds", published 1970-04-21, issued 1970-04-21, assigned to Eli Lilly and Co "Espacenet - Bibliographic data". Archived from the original on 25 September 2022. Retrieved 3 March 2020.
- ^ McPherson EM (2007). "Cefalexin". Pharmaceutical Manufacturing Encyclopedia (3rd ed.). Burlington: Elsevier. p. 915. ISBN 9780815518563. Archived from the original on 8 September 2017.
- ^ Ravina E (2011). The evolution of drug discovery : from traditional medicines to modern drugs (1. Aufl. ed.). Weinheim: Wiley-VCH. p. 267. ISBN 9783527326693. Archived from the original on 8 September 2017.
- ^ Hanlon G, Hodges N (2012). Essential Microbiology for Pharmacy and Pharmaceutical Science. Hoboken: Wiley. p. 140. ISBN 9781118432433. Archived from the original on 8 September 2017.
- ^ World Health Organization (2021). World Health Organization model list of essential medicines: 22nd list (2021). Geneva: World Health Organization. hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Cephalexin - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- ^ "Human Antimicrobial Drug Use Report 2012/2013" (PDF). Public Health Agency of Canada (PHAC). November 2014. Archived (PDF) from the original on 21 March 2015. Retrieved 24 February 2015.
- ^ Australia's Health 2012: The Thirteenth Biennial Health Report of the Australian Institute of Health and Welfare. Australian Institute of Health and Welfare. 2012. p. 408. ISBN 9781742493053. Archived from the original on 8 September 2017.