| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
Chloroacetic acid | |
| Systematic IUPAC name
Chloroethanoic acid | |
| Other names
2-Chloroacetic acid
2-Chloroethanoic acid | |
| Identifiers | |
3D model (JSmol)
|
|
| 3DMet | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.001.072 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
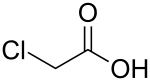
| ClCH2CO2H | |
| Molar mass | 94.49 g·mol−1 |
| Appearance | Colorless or white crystals |
| Density | 1.58 g/cm3 |
| Melting point | 63 °C (145 °F; 336 K) |
| Boiling point | 189.3 °C (372.7 °F; 462.4 K) |
| 85.8 g/(100 mL) (25 °C) | |
| Solubility | Soluble in methanol, acetone, diethyl ether, benzene, chloroform, ethanol |
| log P | 0.22 |
| Vapor pressure | 0.22 hPa |
| Acidity (pKa) | 2.86[1] |
| −48.1×10−6 cm3/mol | |
Refractive index (nD)
|
1.4351 (55 °C) |
| Structure | |
| Monoclinic | |
| Thermochemistry | |
Heat capacity (C)
|
144.02 J/(K·mol) |
Std enthalpy of
formation (ΔfH⦵298) |
−490.1 kJ/mol |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
alkylating agent |
| GHS labelling: | |
  
| |
| Danger | |
| H301, H311, H314, H331, H400 | |
| P260, P261, P264, P270, P271, P273, P280, P301+P310, P301+P330+P331, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P310, P311, P312, P321, P322, P330, P361, P363, P391, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | 126 °C (259 °F; 399 K) |
| 470 °C (878 °F; 743 K) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
76 mg/kg.[2] |
| Safety data sheet (SDS) | External MSDS |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Chloroacetic acid, industrially known as monochloroacetic acid (MCA), is the organochlorine compound with the formula ClCH2CO2H. This carboxylic acid is a useful building block in organic synthesis. It is a colorless solid. Related compounds are dichloroacetic acid and trichloroacetic acid.
- ^ Dippy, J. F. J.; Hughes, S. R. C.; Rozanski, A. (1959). "498. The dissociation constants of some symmetrically disubstituted succinic acids". Journal of the Chemical Society. 1959: 2492–2498. doi:10.1039/JR9590002492.
- ^ Cite error: The named reference
Ullmannwas invoked but never defined (see the help page).
