| |
| Names | |
|---|---|
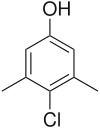
| Preferred IUPAC name
4-Chloro-3,5-dimethylphenol[1] | |
| Other names
para-Chloro-meta-xylenol, PCMX, 4-Chloro-3,5-dimethylphenol
| |
| Identifiers | |
3D model (JSmol)
|
|
| 1862539 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.001.631 |
| EC Number |
|
| KEGG | |
| MeSH | chloroxylenol |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C8H9ClO | |
| Molar mass | 156.61 g·mol−1 |
| Melting point | 115 °C (239 °F; 388 K) |
| Boiling point | 246 °C (475 °F; 519 K) |
| 300 mg/L [2] | |
| Solubility in alcohols | soluble |
| Solubility in Ethers | soluble |
| Solubility in Benzene | soluble |
| log P | 3.377 |
| Acidity (pKa) | 9.76 |
| Basicity (pKb) | 4.24 |
| Pharmacology | |
| D08AE05 (WHO) | |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H302, H315, H317, H319 | |
| P280, P305+P351+P338 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Chloroxylenol, also known as para-chloro-meta-xylenol (PCMX), is a chlorine substituted phenol with a white to off-white appearance and a phenolic odor.
The discovery of chloroxylenol was the result of efforts to produce improved antiseptics that began at the end of the 1800s, when scientists gradually realized that more substituted and more lipophylic phenols are less toxic, less irritant and more powerful.[3] First synthesized in Germany in 1923, it was borne out of the study of coal tar components that began a decade earlier.[2]
- ^ CID 2723 from PubChem
- ^ a b Cite error: The named reference
Ascenziwas invoked but never defined (see the help page). - ^ de Solis NM (1993). Effect of plasmids that confer preservative-resistance on the performance of bacteria in preservative efficacy tests (Doctoral thesis). University College London. p. 31.