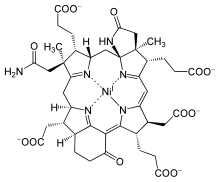
| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| C 42H 51N 6NiO− 13 | |
| Molar mass | 906.58014 |
| Appearance | Yellow solid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
F430 is the cofactor (sometimes called the coenzyme) of the enzyme methyl coenzyme M reductase (MCR).[1][2] MCR catalyzes the reaction EC 2.8.4.1 that releases methane in the final step of methanogenesis:
 |
 |
It is found only in methanogenic Archaea[3] and anaerobic methanotrophic Archaea. It occurs in relatively high concentrations in archaea that are involved in reverse methanogenesis: these can contain up to 7% by weight of the nickel protein.[4]
- ^ Stephen W., Ragdale (2014). "Biochemistry of Methyl-Coenzyme M Reductase: The Nickel Metalloenzyme that Catalyzes the Final Step in Synthesis and the First Step in Anaerobic Oxidation of the Greenhouse Gas Methane". In Peter M.H. Kroneck and Martha E. Sosa Torres (ed.). The Metal-Driven Biogeochemistry of Gaseous Compounds in the Environment. Metal Ions in Life Sciences. Vol. 14. Springer. pp. 125–145. doi:10.1007/978-94-017-9269-1_6. ISBN 978-94-017-9268-4. PMID 25416393.
- ^ Hofer, Ursula (2016). "Masters of methane". Nature Reviews Microbiology. 14 (12): 727. doi:10.1038/nrmicro.2016.170. PMID 27818502. S2CID 35175663.
- ^ Thauer RK (1998). "Biochemistry of Methanogenesis: a Tribute to Marjory Stephenson". Microbiology. 144 (9): 2377–2406. doi:10.1099/00221287-144-9-2377. PMID 9782487.
- ^ Krüger M, Meyerdierks A, Glöckner FO, et al. (December 2003). "A conspicuous nickel protein in microbial mats that oxidize methane anaerobically". Nature. 426 (6968): 878–81. Bibcode:2003Natur.426..878K. doi:10.1038/nature02207. PMID 14685246. S2CID 4383740.