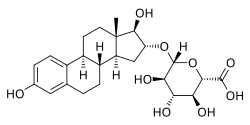
 Estriol 16α-glucuronide, the primary active component in conjugated estriol (estriol glucuronides constitute about 35 to 46% of total content). | |
 Estriol 3-sulfate, the second major active component in conjugated estriol (constitutes about 15 to 22% of total content). | |
| Combination of | |
|---|---|
| Estriol glucuronide | Estrogen |
| Estriol sulfate | Estrogen |
| Estriol sulfate glucuronide | Estrogen |
| Clinical data | |
| Trade names | Progynon, Emmenin |
| Other names | Conjugated human estriol; Conjugated oestriol; Conjugated human oestriol; CE3; CHE3; Conjugated human estrogens; Conjugated human oestrogens; CHEs |
| Routes of administration | By mouth |
| Identifiers | |
| CAS Number | |
| UNII | |
Conjugated estriol, sold under the brand names Progynon and Emmenin, is an estrogen medication which was previously used for estrogen-type indications such as the treatment of menopausal symptoms in women. The term specifically refers to formulations of estriol conjugates which were manufactured from the estrogen-rich urine of pregnant women and were used as medications in the 1920s and 1930s. Conjugated estriol is analogous to and was superseded by conjugated estrogens (brand name Premarin), which is manufactured from the urine of pregnant mares. Conjugated estriol was among the first forms of pharmaceutical estrogen to be used in medicine. It was taken by mouth.
The main components of conjugated estriol are estriol glucuronides and to a lesser extent estriol sulfates. Estrogen glucuronides can be deglucuronidated into the corresponding free estrogens by β-glucuronidase in tissues that express this enzyme, such as the mammary gland, liver, and kidney, among others.[1] Likewise, estrogen sulfates can be desulfated into the corresponding free estrogens by steroid sulfatase in tissues that express this enzyme.[2] Consequently, estrogen conjugates have estrogenic activity via conversion into unconjugated estrogens.[1]
- ^ a b Zhu BT, Conney AH (January 1998). "Functional role of estrogen metabolism in target cells: review and perspectives". Carcinogenesis. 19 (1): 1–27. doi:10.1093/carcin/19.1.1. PMID 9472688.
- ^ Kuhl H (August 2005). "Pharmacology of estrogens and progestogens: influence of different routes of administration". Climacteric. 8 (Suppl 1): 3–63. doi:10.1080/13697130500148875. PMID 16112947. S2CID 24616324.