 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Tafinlar |
| Other names | GSK-2118436 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a613038 |
| License data |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.215.965 |
| Chemical and physical data | |
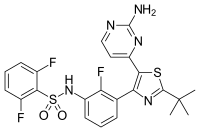
| Formula | C23H20F3N5O2S2 |
| Molar mass | 519.56 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Dabrafenib, sold under the brand name Tafinlar among others, is an anti-cancer medication used for the treatment of cancers associated with a mutated version of the gene BRAF.[2] Dabrafenib acts as an inhibitor of the associated enzyme B-Raf, which plays a role in the regulation of cell growth.
The most common side effects include papilloma (warts), headache, nausea, vomiting, hyperkeratosis (thickening and toughening of the skin), hair loss, rash, joint pain, fever and tiredness.[3] When taken in combination with trametinib, the most common side effects include fever, tiredness, nausea, chills, headache, diarrhea, vomiting, joint pain and rash.[3]
Dabrafenib was approved for medical use in the United States in May 2013,[7] and in the European Union in August 2013.[3]
- ^ "Product monograph brand safety updates". Health Canada. February 2024. Retrieved March 24, 2024.
- ^ a b "Tafinlar- dabrafenib capsule". DailyMed. U.S. National Library of Medicine. June 22, 2022. Archived from the original on January 27, 2023. Retrieved January 27, 2023.
- ^ a b c d Cite error: The named reference
Tafinlar EPARwas invoked but never defined (see the help page). - ^ Cite error: The named reference
Finlee EPARwas invoked but never defined (see the help page). - ^ "Tafinlar Product information". Union Register of medicinal products. August 29, 2013. Archived from the original on January 11, 2023. Retrieved December 11, 2023.
- ^ "Finlee Product information". Union Register of medicinal products. November 16, 2023. Archived from the original on November 26, 2023. Retrieved December 11, 2023.
- ^ Cite error: The named reference
Tafinlar FDA approval packagewas invoked but never defined (see the help page).