
| |
| |
| Names | |
|---|---|
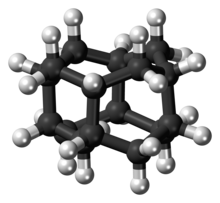
| IUPAC name
Pentacyclo[7.3.1.14,12.02,7.06,11]tetradecane
| |
| Other names
Congressane, diadamantane, decahydro-3,5,1,7-[1,2,3,4]butanetetraylnaphthalene
| |
| Identifiers | |
3D model (JSmol)
|
|
| 1904934 | |
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C14H20 | |
| Molar mass | 188.314 g·mol−1 |
| Appearance | solid white crystal |
| Density | 1.092 ± 0.06 g cm−3 |
| Melting point | 244.73 °C (472.51 °F; 517.88 K) |
| Boiling point | 269.5 ± 7.0 °C |
| sparingly soluble (8.1 x 10−4 g L−1) | |
| Solubility in diethyl ether | soluble |
| log P | 5.556 ± 0.228 |
| Vapor pressure | 0.0120 Torr |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
irritant, acute toxicity to aquatic life |
| NFPA 704 (fire diamond) | |
| Flash point | 98.1 °C (208.6 °F; 371.2 K) |
| Safety data sheet (SDS) | External MSDS |
| Structure | |
| D3d | |
| 0 D | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Diamantane (also called congressane) is an organic compound that is a member of the diamondoids. These are cage hydrocarbons with structures similar to a subunit of the diamond lattice. It is a colorless solid that has been a topic of research since its discovery in oil and separation from deep natural gas condensates. Diamondoids such as diamantane exhibit unusual properties, including low surface energies, high densities, high hydrophobicities, and resistance to oxidation.[1]
- ^ Schreiner, Peter; Fokin, Andrey; Fokina, Natalie; Tkachenko, Boryslav; Dahl, Jeremy; Carlson, Robert (2011). "Synthesis of Diamondoid Carboxylic Acids". Synthesis. 2012 (2): 259. doi:10.1055/s-0031-1289617.
