
| |
| |
| Names | |
|---|---|
| IUPAC name
Diethyl diazenedicarboxylate
| |
| Systematic IUPAC name
Ethyl N-ethoxycarbonyliminocarbamate | |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.016.202 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C6H10N2O4 | |
| Molar mass | 174.156 g·mol−1 |
| Appearance | Orange to red to orange liquid[2] |
| Density | 1.11 g/cm3[3] |
| Melting point | 6 °C (43 °F; 279 K)[4] |
| Boiling point | 104.5 °C (220.1 °F; 377.6 K) at 12 mm Hg[3] |
Refractive index (nD)
|
1.420 (20 °C)[3] |
| Hazards | |
| GHS labelling: | |
 
| |
| Danger | |
| H240, H302, H312, H315, H319, H332, H335 | |
| P210, P220, P234, P261, P264, P270, P271, P280, P301+P312, P302+P352, P304+P312, P304+P340, P305+P351+P338, P312, P321, P322, P330, P332+P313, P337+P313, P362, P363, P370+P378, P370+P380+P375, P403+P233, P403+P235, P405, P411, P420, P501 | |
| Flash point | 85 °C (185 °F; 358 K)[5] |
| Safety data sheet (SDS) | External MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
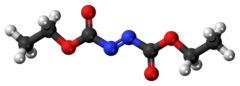
Diethyl azodicarboxylate, conventionally abbreviated as DEAD and sometimes as DEADCAT,[6][7] is an organic compound with the structural formula CH3CH2−O−C(=O)−N=N−C(=O)−O−CH2CH3. Its molecular structure consists of a central azo functional group, RN=NR, flanked by two ethyl ester groups. This orange-red liquid is a valuable reagent but also quite dangerous and explodes upon heating. Therefore, commercial shipment of pure diethyl azodicarboxylate is prohibited in the United States and is carried out either in solution or on polystyrene particles.
DEAD is an aza-dienophile and an efficient dehydrogenating agent, converting alcohols to aldehydes, thiols to disulfides and hydrazo groups to azo groups; it is also a good electron acceptor. While DEAD is used in numerous chemical reactions it is mostly known as a key component of the Mitsunobu reaction, a common strategy for the preparation of an amine, azide, ether, thioether, or ester from the corresponding alcohol.[8] It is used in the synthesis of various natural products and pharmaceuticals such as zidovudine, an AIDS drug; FdUMP, a potent antitumor agent; and procarbazine, a chemotherapy drug.
- ^ Diethyl azodiformate. Webbook.nist.gov (1972-07-28). Retrieved on 2011-03-15.
- ^ Cite error: The named reference
msdswas invoked but never defined (see the help page). - ^ a b c Cite error: The named reference
b1was invoked but never defined (see the help page). - ^ Cite error: The named reference
s1was invoked but never defined (see the help page). - ^ L19348 Diethyl azodicarboxylate, 97% – Alfa Aesar – A Johnson Matthey Company. Alfa.com (1972-07-28). Retrieved on 2011-03-15.
- ^ Caroline Cooper (23 July 2010). Organic Chemist's Desk Reference. CRC Press. pp. 109–. ISBN 978-1-4398-1164-1. Retrieved 12 March 2011.
- ^ DEAD is sometimes used for another chemical, diethyl acetylenedicarboxylate. P. N. Preston (1980). Benzimidazoles and congeneric tricyclic compounds. John Wiley and Sons. pp. 475–. ISBN 978-0-471-08189-0. Retrieved 12 March 2011.
- ^ Barbara., Czakó (2009). Strategic applications of named reactions in organic synthesis: background and detailed mechanisms ; 250 named reactions. Elsevier. ISBN 978-0-12-369483-6. OCLC 634820219.