 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Unisom, Vicks Formula 44 (in combination with Dextromethorphan), others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682537 |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Oral: 24.7%[1] Intranasal: 70.8%[1] |
| Metabolism | Hepatic (CYP2D6, CYP1A2, CYP2C9)[2] |
| Elimination half-life | 10–12 hours (range 7–15 hours)[2][3][4] |
| Excretion | Urine (60%), feces (40%)[5] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.006.742 |
| Chemical and physical data | |
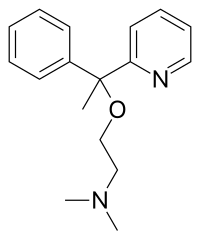
| Formula | C17H22N2O |
| Molar mass | 270.376 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Doxylamine is an antihistamine medication used to treat insomnia and allergies, and—in combination with pyridoxine (vitamin B6)—to treat morning sickness in pregnant women. It is available over-the-counter and is typically sold under such brand names as Equate or Unisom, among others; and it is used in nighttime cold medicines (e.g., NyQuil) and pain medications containing acetaminophen and/or codeine to help with sleep. The medication is delivered chemically by the salt doxylamine succinate and is taken by mouth. Doxylamine and other first-generation antihistamines are the most widely used sleep medications in the world.[6] Typical side effects of doxylamine (at recommended doses) include dizziness, drowsiness, grogginess, and dry mouth, among others.[7][4]
As an antihistamine, doxylamine is an inverse agonist of the histamine H1 receptor. As a first-generation antihistamine, it typically crosses the blood–brain barrier into the brain, thereby producing a suite of sedative and hypnotic effects that are mediated by the central nervous system. (N.b.: An agonist is a molecule that activates certain receptors (i.e., specific cellular proteins) in a cell to produce a specific pharmacological response, causing the cell to modify its activity—while an inverse agonist targets the same receptors as those of a given agonist, but causes a response opposite to that caused by the agonist. An antagonist blocks the action of a given agonist.)
Doxylamine is also a potent anticholinergic, meaning that it causes delirum at high doses—i.e., at much higher doses than recommended.[8] (Specifically it is an antagonist of the muscarinic acetylcholine receptors M1 through M5.) These sedative and deliriant effects have in some cases led to using the drug recreationally. Doxylamine was first described in 1948 or 1949.[9]
- ^ a b Cite error: The named reference
pmid12214324was invoked but never defined (see the help page). - ^ a b Cite error: The named reference
KrygerRoth2010was invoked but never defined (see the help page). - ^ Cite error: The named reference
pmid29671128was invoked but never defined (see the help page). - ^ a b Cite error: The named reference
pmid27057416was invoked but never defined (see the help page). - ^ "New Zealand Datasheet: Doxylamine Succinate" (PDF). Medsafe, New Zealand Medicines and Medical Devices Safety Authority. 16 July 2008. Archived from the original on 22 March 2016.
- ^ Cite error: The named reference
SimonsSimons2011was invoked but never defined (see the help page). - ^ Neubauer DN (August 2007). "The evolution and development of insomnia pharmacotherapies". Journal of Clinical Sleep Medicine. 3 (5 Suppl): S11–S15. doi:10.5664/jcsm.26930. PMC 1978321. PMID 17824496.
- ^ "Doxylamine - PsychonautWiki".
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 546. ISBN 9783527607495.