
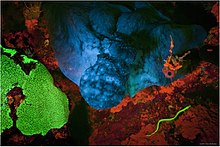

Fluorescence is one of two kinds of emission of light by a substance that has absorbed light or other electromagnetic radiation. When exposed to ultraviolet radiation, many substances will glow (fluoresce) with colored visible light. The color of the light emitted depends on the chemical composition of the substance. Fluorescent materials generally cease to glow nearly immediately when the radiation source stops. This distinguishes them from the other type of light emission, phosphorescence. Phosphorescent materials continue to emit light for some time after the radiation stops.
Fluorescence occurs when a photon of the incoming radiation is absorbed by an orbital electron in a molecule of the material, exciting it to a higher energy level. The electron then returns to its former energy level by losing energy, emitting another photon of a different frequency, which is the fluorescent glow.
Fluorescence is a form of luminescence. In nearly all cases, the emitted light has a longer wavelength, and therefore a lower photon energy, than the absorbed radiation. The most common example occurs when the absorbed radiation is in the ultraviolet region of the electromagnetic spectrum (invisible to the human eye), while the emitted light is in the visible region. This gives the fluorescent substance a distinct color that is best seen when it has been exposed to UV light, making it appear to glow in the dark. However, any light of a shorter wavelength may cause a material to fluoresce at a longer wavelength. Fluorescent materials may also be excited by certain wavelengths of visible light, which masks the glow, yet their colors may appear bright and intensified. Other fluorescent materials emit their light in the infrared or even the ultraviolet regions of the spectrum.
Fluorescence has many practical applications, including mineralogy, gemology, medicine, chemical sensors (fluorescence spectroscopy), fluorescent labelling, dyes, biological detectors, cosmic-ray detection, vacuum fluorescent displays, and cathode-ray tubes. Its most common everyday application is in (gas-discharge) fluorescent lamps and LED lamps, in which fluorescent coatings convert UV or blue light into longer-wavelengths resulting in white light which can even appear indistinguishable from that of the traditional but energy-inefficient incandescent lamp. Fluorescence also occurs frequently in nature in some minerals and in many biological forms across all kingdoms of life. The latter may be referred to as biofluorescence, indicating that the fluorophore is part of or is extracted from a living organism (rather than an inorganic dye or stain). But since fluorescence is due to a specific chemical, which can also be synthesized artificially in most cases, it is sufficient to describe the substance itself as fluorescent.