 | |
| Clinical data | |
|---|---|
| Trade names | Glurenorm |
| AHFS/Drugs.com | International Drug Names |
| Pregnancy category |
|
| Routes of administration | Oral (tablets) |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | High (Tmax = 2–3 hours) |
| Metabolism | Extensive hepatic |
| Onset of action | 1–1.5 hours |
| Excretion | Biliary (95%), renal (5%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.046.770 |
| Chemical and physical data | |
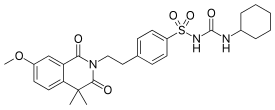
| Formula | C27H33N3O6S |
| Molar mass | 527.64 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Gliquidone (INN, sold under the trade name Glurenorm) is an anti-diabetic medication in the sulfonylurea class.[1] It is classified as a second-generation sulfonylurea. It is used in the treatment of diabetes mellitus type 2. It is marketed by the pharmaceutical company Boehringer Ingelheim (Germany).