| Glyceraldehyde 3-phosphate dehydrogenase, NAD binding domain | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 determinants of enzyme thermostability observed in the molecular structure of thermus aquaticus d-glyceraldehyde-3-phosphate dehydrogenase at 2.5 angstroms resolution | |||||||||
| Identifiers | |||||||||
| Symbol | Gp_dh_N | ||||||||
| Pfam | PF00044 | ||||||||
| Pfam clan | CL0063 | ||||||||
| InterPro | IPR020828 | ||||||||
| PROSITE | PDOC00069 | ||||||||
| SCOP2 | 1gd1 / SCOPe / SUPFAM | ||||||||
| |||||||||
| Glyceraldehyde 3-phosphate dehydrogenase, C-terminal domain | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 crystal structure of glyceraldehyde-3-phosphate dehydrogenase from pyrococcus horikoshii ot3 | |||||||||
| Identifiers | |||||||||
| Symbol | Gp_dh_C | ||||||||
| Pfam | PF02800 | ||||||||
| Pfam clan | CL0139 | ||||||||
| InterPro | IPR020829 | ||||||||
| PROSITE | PDOC00069 | ||||||||
| SCOP2 | 1gd1 / SCOPe / SUPFAM | ||||||||
| |||||||||
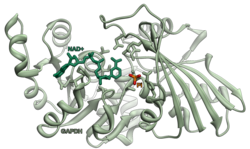
Glyceraldehyde 3-phosphate dehydrogenase (abbreviated GAPDH) (EC 1.2.1.12) is an enzyme of about 37kDa that catalyzes the sixth step of glycolysis and thus serves to break down glucose for energy and carbon molecules. In addition to this long established metabolic function, GAPDH has recently been implicated in several non-metabolic processes, including transcription activation, initiation of apoptosis,[4] ER-to-Golgi vesicle shuttling, and fast axonal, or axoplasmic transport.[5] In sperm, a testis-specific isoenzyme GAPDHS is expressed.
- ^ a b c GRCh38: Ensembl release 89: ENSG00000111640 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Tarze A, Deniaud A, Le Bras M, Maillier E, Molle D, Larochette N, Zamzami N, Jan G, Kroemer G, Brenner C (April 2007). "GAPDH, a novel regulator of the pro-apoptotic mitochondrial membrane permeabilization". Oncogene. 26 (18): 2606–2620. doi:10.1038/sj.onc.1210074. PMID 17072346. S2CID 20291542.
- ^ Zala D, Hinckelmann MV, Yu H, Lyra da Cunha MM, Liot G, Cordelières FP, Marco S, Saudou F (January 2013). "Vesicular glycolysis provides on-board energy for fast axonal transport". Cell. 152 (3): 479–491. doi:10.1016/j.cell.2012.12.029. PMID 23374344.