
- Stage 1 (3.85–2.45 Ga): Practically no O2 in the atmosphere. The oceans were also largely anoxic – with the possible exception of O2 in the shallow oceans.
- Stage 2 (2.45–1.85 Ga): O2 produced, rising to values of 0.02 and 0.04 atm, but absorbed in oceans and seabed rock.
- Stage 3 (1.85–0.85 Ga): O2 starts to gas out of the oceans, but is absorbed by land surfaces. No significant change in oxygen level.
- Stages 4 and 5 (0.85 Ga – present): Other O2 reservoirs filled; gas accumulates in atmosphere.[1]
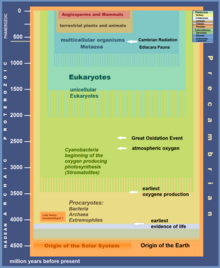
The Great Oxidation Event (GOE) or Great Oxygenation Event, also called the Oxygen Catastrophe, Oxygen Revolution, Oxygen Crisis or Oxygen Holocaust,[2] was a time interval during the Earth's Paleoproterozoic era when the Earth's atmosphere and shallow seas first experienced a rise in the concentration of free oxygen.[3] This began approximately 2.460–2.426 Ga (billion years) ago during the Siderian period and ended approximately 2.060 Ga ago during the Rhyacian.[4] Geological, isotopic and chemical evidence suggests that biologically produced molecular oxygen (dioxygen or O2) started to accumulate in the Archean prebiotic atmosphere due to microbial photosynthesis, and eventually changed it from a weakly reducing atmosphere practically devoid of oxygen into an oxidizing one containing abundant free oxygen,[5] with oxygen levels being as high as 10% of modern atmospheric level by the end of the GOE.[6]
The appearance of highly reactive free oxygen, which can oxidize organic compounds (especially genetic materials) and thus is toxic to the then-mostly anaerobic biosphere, may have caused the extinction/extirpation of many early organisms on Earth – mostly archaeal colonies that used retinal to use green-spectrum light energy and power a form of anoxygenic photosynthesis (see Purple Earth hypothesis). Although the event is inferred to have constituted a mass extinction,[7] due in part to the great difficulty in surveying microscopic organisms' abundances, and in part to the extreme age of fossil remains from that time, the Great Oxidation Event is typically not counted among conventional lists of "great extinctions", which are implicitly limited to the Phanerozoic eon. In any case, isotope geochemistry data from sulfate minerals have been interpreted to indicate a decrease in the size of the biosphere of >80% associated with changes in nutrient supplies at the end of the GOE.[8]
The GOE is inferred to have been caused by cyanobacteria, which evolved chlorophyll-based photosynthesis that releases dioxygen as a byproduct of water photolysis. The continually produced oxygen eventually depleted all the surface reducing capacity from ferrous iron, sulfur, hydrogen sulfide and atmospheric methane over nearly a billion years. The oxidative environmental change, compounded by a global glaciation, devastated the microbial mats around the Earth's surface. The subsequent adaptation of surviving archaea via symbiogenesis with aerobic proteobacteria (which went endosymbiont and became mitochondria) may have led to the rise of eukaryotic organisms and the subsequent evolution of multicellular life-forms.[9][10][11]
- ^ Holland, Heinrich D. (19 May 2006). "The oxygenation of the atmosphere and oceans". Philosophical Transactions of the Royal Society: Biological Sciences. 361 (1470): 903–915. doi:10.1098/rstb.2006.1838. PMC 1578726. PMID 16754606.
- ^ Margulis, Lynn; Sagan, Dorion (1986). "Chapter 6, "The Oxygen Holocaust"". Microcosmos: Four Billion Years of Microbial Evolution. California: University of California Press. p. 99. ISBN 9780520210646.
- ^ Lyons, Timothy W.; Reinhard, Christopher T.; Planavsky, Noah J. (February 2014). "The rise of oxygen in Earth's early ocean and atmosphere". Nature. 506 (7488): 307–315. Bibcode:2014Natur.506..307L. doi:10.1038/nature13068. PMID 24553238. S2CID 4443958.
- ^ Gumsley, Ashley P.; Chamberlain, Kevin R.; Bleeker, Wouter; Söderlund, Ulf; De Kock, Michiel O.; Larsson, Emilie R.; Bekker, Andrey (6 February 2017). "Timing and tempo of the Great Oxidation Event". Proceedings of the National Academy of Sciences of the United States of America. 114 (8): 1811–1816. Bibcode:2017PNAS..114.1811G. doi:10.1073/pnas.1608824114. ISSN 0027-8424. PMC 5338422. PMID 28167763.
- ^ Sosa Torres, Martha E.; Saucedo-Vázquez, Juan P.; Kroneck, Peter M.H. (2015). "The Magic of Dioxygen". In Kroneck, Peter M.H.; Sosa Torres, Martha E. (eds.). Sustaining Life on Planet Earth: Metalloenzymes Mastering Dioxygen and Other Chewy Gases. Metal Ions in Life Sciences volume 15. Vol. 15. Springer. pp. 1–12. doi:10.1007/978-3-319-12415-5_1. ISBN 978-3-319-12414-8. PMID 25707464.
- ^ Ossa Ossa, Frantz; Spangenberg, Jorge E.; Bekker, Andrey; König, Stephan; Stüeken, Eva E.; Hofmann, Axel; et al. (15 September 2022). "Moderate levels of oxygenation during the late stage of Earth's Great Oxidation Event". Earth and Planetary Science Letters. 594: 117716. Bibcode:2022E&PSL.59417716O. doi:10.1016/j.epsl.2022.117716. hdl:10481/78482.
- ^ Plait, Phil (28 July 2014). "Poisoned Planet". Slate. Retrieved 8 July 2019.
- ^ Hodgskiss, Malcolm S. W.; Crockford, Peter W.; Peng, Yongbo; Wing, Boswell A.; Horner, Tristan J. (27 August 2019). "A productivity collapse to end Earth's Great Oxidation". Proceedings of the National Academy of Sciences of the United States of America. 116 (35): 17207–17212. Bibcode:2019PNAS..11617207H. doi:10.1073/pnas.1900325116. ISSN 0027-8424. PMC 6717284. PMID 31405980.
- ^ Schirrmeister, Bettina E.; de Vos, Jurriaan M.; Antonelli, Alexandre; Bagheri, Homayoun C. (29 January 2013). "Evolution of multicellularity coincided with increased diversification of cyanobacteria and the Great Oxidation Event". Proceedings of the National Academy of Sciences of the United States of America. 110 (5): 1791–1796. Bibcode:2013PNAS..110.1791S. doi:10.1073/pnas.1209927110. PMC 3562814. PMID 23319632.
- "Great Oxidation Event: More oxygen through multicellularity". ScienceDaily (Press release). 17 January 2013.
- ^ Crockford, Peter W.; Kunzmann, Marcus; Bekker, Andrey; Hayles, Justin; Bao, Huiming; Halverson, Galen P.; et al. (20 May 2019). "Claypool continued: Extending the isotopic record of sedimentary sulfate". Chemical Geology. 513: 200–225. Bibcode:2019ChGeo.513..200C. doi:10.1016/j.chemgeo.2019.02.030. ISSN 0009-2541.
- ^ Crockford, Peter W.; bar On, Yinon M.; Ward, Luce M.; Milo, Ron; Halevy, Itay (November 2023). "The geologic history of primary productivity". Current Biology. 33 (21): 4741–4750.e5. Bibcode:2023CBio...33E4741C. doi:10.1016/j.cub.2023.09.040. PMID 37827153.